How is the drug name pronounced?
Afamitresgene autoleucel: A-fam-ih-TRES-jeen AW-toh-LOO-sel
Tecelra: te-SEL-ruh
What cancer(s) does this drug treat?
Synovial Sarcoma
Tecelra is approved for:
- Patients with synovial sarcoma that cannot be removed or has spread (metastatic), who have previously been treated with chemotherapy, test positive for HLA-A*02:01P, -A*02:02P, -A*02:03P, or -A*02:06P, and whose cancer cells contain the MAGE-A4 antigen.
Limitations of Use
Age: The safety and efficacy of Tecelra have not been established in patients under 18 years of age.
Fertility/Pregnancy/Breastfeeding: The effects of Tecelra on female and male fertility are not known. Tecelra is not recommended for patients who are pregnant, and pregnancy after treatment should be discussed with the treating physician. The effects of Tecelra on a breastfed infant are also unknown, and the risks and benefits of breastfeeding should be discussed with a physician.
Effects on the ability to drive and use machines: Patients are advised to refrain from driving, engaging in hazardous occupations or activities, and operating heavy machinery for at least 4 weeks after receiving Tecelra.
HLA Variants: Tecelra is not for use in patients who test positive for one or two copies of HLA-A*02:05P.
What type of immunotherapy is this?
- TCR-engineered T cell therapy
How does this drug work?
Target:
- MAGE-A4
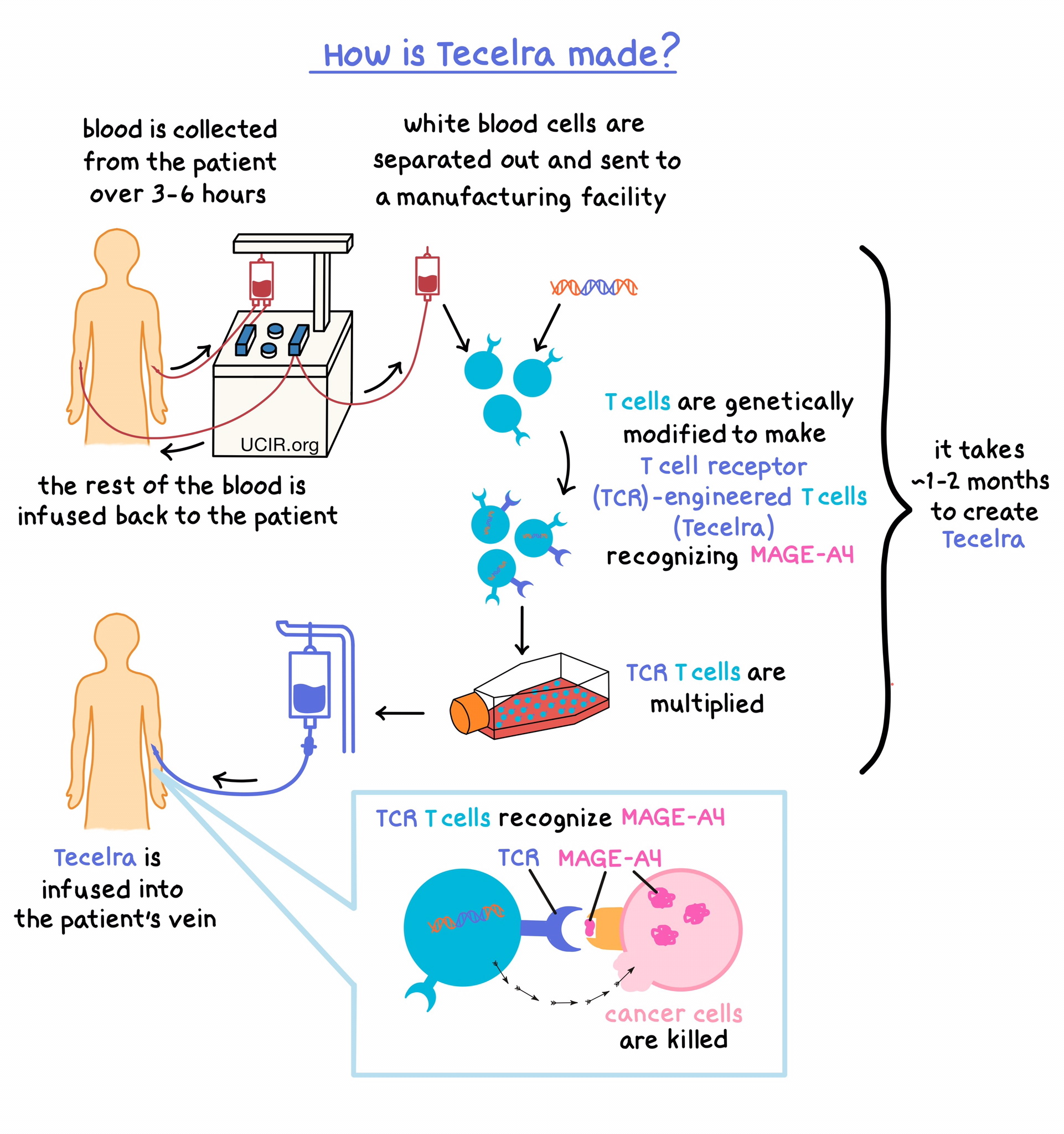
Tecelra is made from the patient’s own T cells (a type of white blood cell). In order to make Tecelra, blood is collected from the patient’s vein in a process called leukapheresis. During this process, the white blood cells (including T cells) are separated from the collected blood, and the rest of the blood is returned to the patient. This process usually takes 3 to 6 hours and may need to be repeated. The collected white blood cells are then sent to a specialized manufacturing facility where the patient’s T cells are genetically modified in such a way that they make a T cell receptor that specifically recognizes the MAGE-A4 antigen that is present in the patient’s cancer cells. The modified T cells (“TCR T cells”) are then multiplied to create millions of TCR T cells. The multiplied TCR T cell product is then delivered back to the patient through a tube in the vein. The entire process, from leukapheresis to treatment administration, takes about 6 weeks.
Once inside the body, the TCR molecules on the surfaces of the modified TCR T cells can attach to a complex of proteins on the surface of the cancer cells representing the MAGE-A4 antigen. When a TCR attaches to the complex, the T cells recognize the cancer cells and kill them.
How is this drug given to the patient?
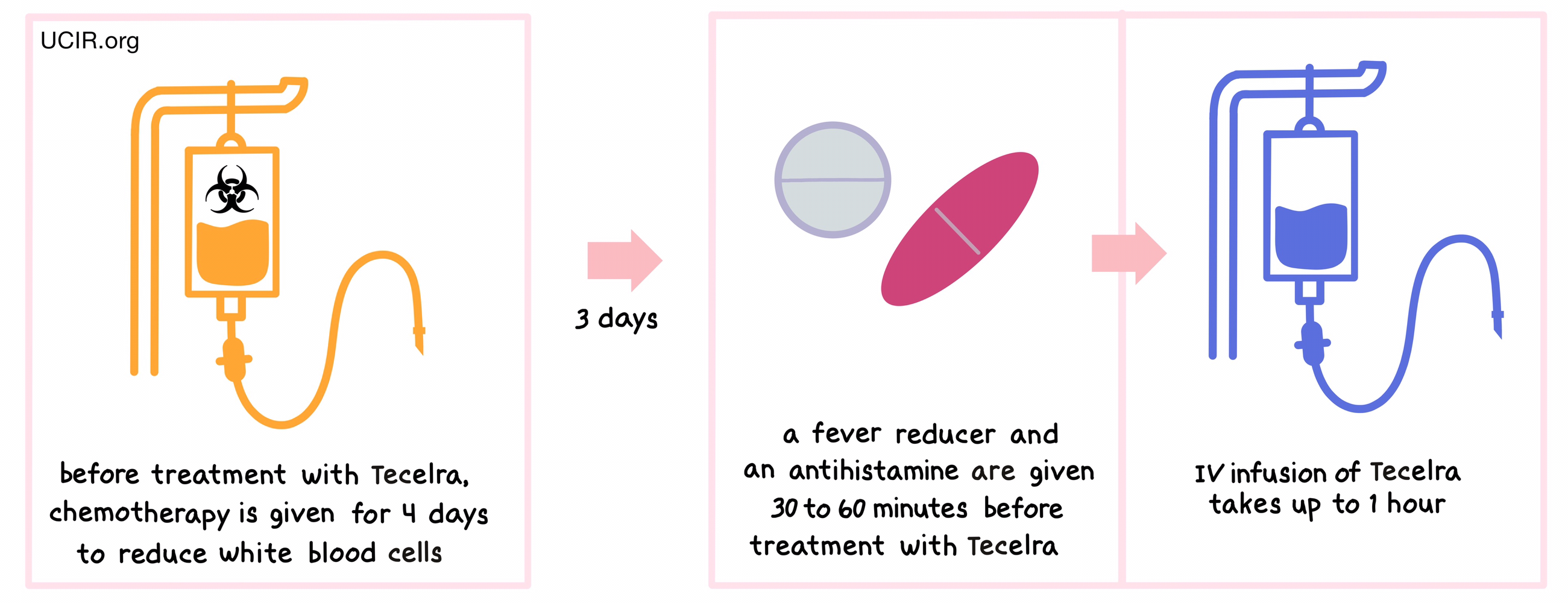
One week prior to receiving Tecelra, patients are treated with a 4-day course of chemotherapy (cyclophosphamide and fludarabine) to reduce the number of white blood cells in the patient’s blood. This is done to make room for the TCR-engineered T cells and ensure that they have the space and resources to survive in the patient’s blood. On the day of the Tecelra administration, patients are treated with an antihistamine and acetaminophen (fever reducer) 30-60 minutes prior to the infusion to reduce the risk of a hypersensitivity (allergic) reaction to the infusion. Tecelra is then administered intravenously (i.v., through a tube into a vein) over the course of up to 1 hour.
Patients are monitored at the healthcare facility for at least 7 days, and then periodically for 4 weeks following treatment with Tecelra. Patients should plan to stay close to the treatment location for the duration of this monitoring period. Patients should refrain from driving or participating in hazardous activities for at least 4 weeks following treatment with Tecelra.
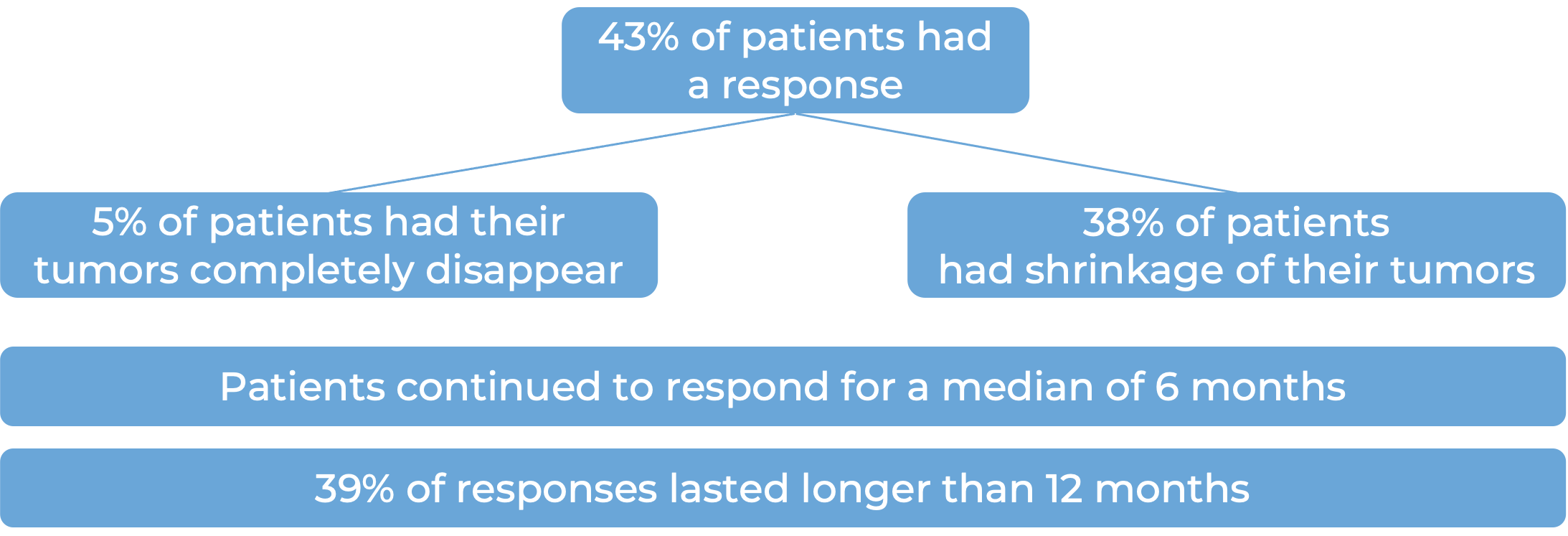
What are the observed clinical results?
It is important to keep in mind that each patient’s actual outcome is individual and may be different from the results found in the clinical studies. In addition, with immunotherapy, sometimes it takes several months for responses to be observed.
Synovial Sarcoma
In a clinical trial, 44 patients with synovial sarcoma that could not be removed or had spread (metastatic), who tested positive for HLA-A*02:01P, HLA-A*02:02P, HLA-A*02:03P, or HLA-A*02:06P, who had received prior chemotherapy with either doxorubicin and/or ifosfamide, and whose tumors contained the MAGE-A4 cancer antigen were treated with Tecelra.
What are the potential side effects?
The most common side effects associated with Tecelra are cytokine release syndrome, nausea, vomiting, fatigue, infections, fever, constipation, shortness of breath, abdominal pain, chest pain (not related to the heart), decreased appetite, increased heart rate, back pain, low blood pressure, diarrhea, swelling, and abnormal blood results.
Some side effects, such as cytokine release syndrome (CRS), immune effector cell-associated neurotoxicity syndrome (ICANS), low blood cell counts (cytopenias), infections, development of other cancers, or hypersensitivity (allergic) reactions to Tecelra may be severe or life-threatening. Patients must be monitored daily at their healthcare facility for at least 7 days for signs and symptoms of severe side effects, and periodically for at least 4 weeks. Patients should remain within proximity of a healthcare facility for at least the 4 weeks following treatment. Patients and caregivers receive careful instructions to monitor for signs and symptoms related to these side effects, and the conditions are managed by the healthcare provider.
Cytokine release syndrome (CRS)
CRS is caused by a widespread release of molecules called cytokines, which are involved in inflammation and can affect the function of various organs. Cytokines may be released by the TCR-engineered T cells in or by other immune cells in the patient’s body. Symptoms of CRS include high fever, fast heart rate, low blood pressure, nausea, vomiting, and headache. CRS typically occurs between 1 and 5 days after infusion, and the healthcare provider should be immediately notified if any symptoms occur.
Immune effector cell-associated neurotoxicity syndrome (ICANS)
Some of the cytokines released during CRS can result in disruption of the blood–brain barrier, leading to the development of neurological toxicities. Symptoms of ICANS include confusion, altered or decreased consciousness, tremors, seizures, headache, loss of balance, and difficulty speaking and understanding.
Serious infections
The lymphodepleting chemotherapy used prior to treatment with TCR-engineered T cells in Tecelra can lead to low levels of various immune cells for long periods of time, which increases the risk of serious, potentially life-threatening infections, including reactivation of viruses that were previously present, but inactive (dormant) in the patient.
Development of other cancers
Patients treated with Tecelra may be at risk for developing new cancers or experiencing recurrence of their existing cancer.
Hypersensitivity reactions
Serious hypersensitivity reactions, including anaphylaxis, may occur during the infusion of Tecelra.
Interference with lab tests for HIV infection
Some lab tests for HIV infection may yield false-positive results in patients who have received Tecelra.
For a more complete list of possible side effects, see the full prescribing information.
Additional Information
Manufacturer
Adaptimmune, LLC
Approval
FDA
Link to drug website
Last update: October 28, 2025


