How is the drug name pronounced?
Amivantamab-vmjw: A-mih-VAN-tuh-mab
Rybrevant: RY-breh-vant
What cancer(s) does this drug treat?
Advanced non-small cell lung cancer
Rybrevant is approved for:
- Patients with non-small cell lung cancer that has grown or spread to other parts of the body on, or after treatment with platinum-based chemotherapy, and tests positive for Epidermal Growth Factor Receptor (EGFR) exon 20 insertion mutations.
Limitations of use:
Age: The safety and efficacy of Rybrevant has not been established in patients under 18 years of age.
Pregnancy/Breastfeeding: Rybrevant may cause harm to the fetus and is not recommended for use during pregnancy. Women are advised to use contraception during treatment with Rybrevant and for at least 3 months after the last dose of Rybrevant. The risks associated with Rybrevant during breastfeeding are not known and cannot be ruled out. Due to the potential for serious side effects in the breastfed child, women are advised not to breastfeed during treatment with Rybrevant and for at least 3 months after the last dose of Rybrevant.
What type of immunotherapy is this?
- Cell growth inhibitor
How does this drug work?
Targets:
- EGFR (epidermal growth factor receptor)
- MET (the hepatocyte growth factor receptor)
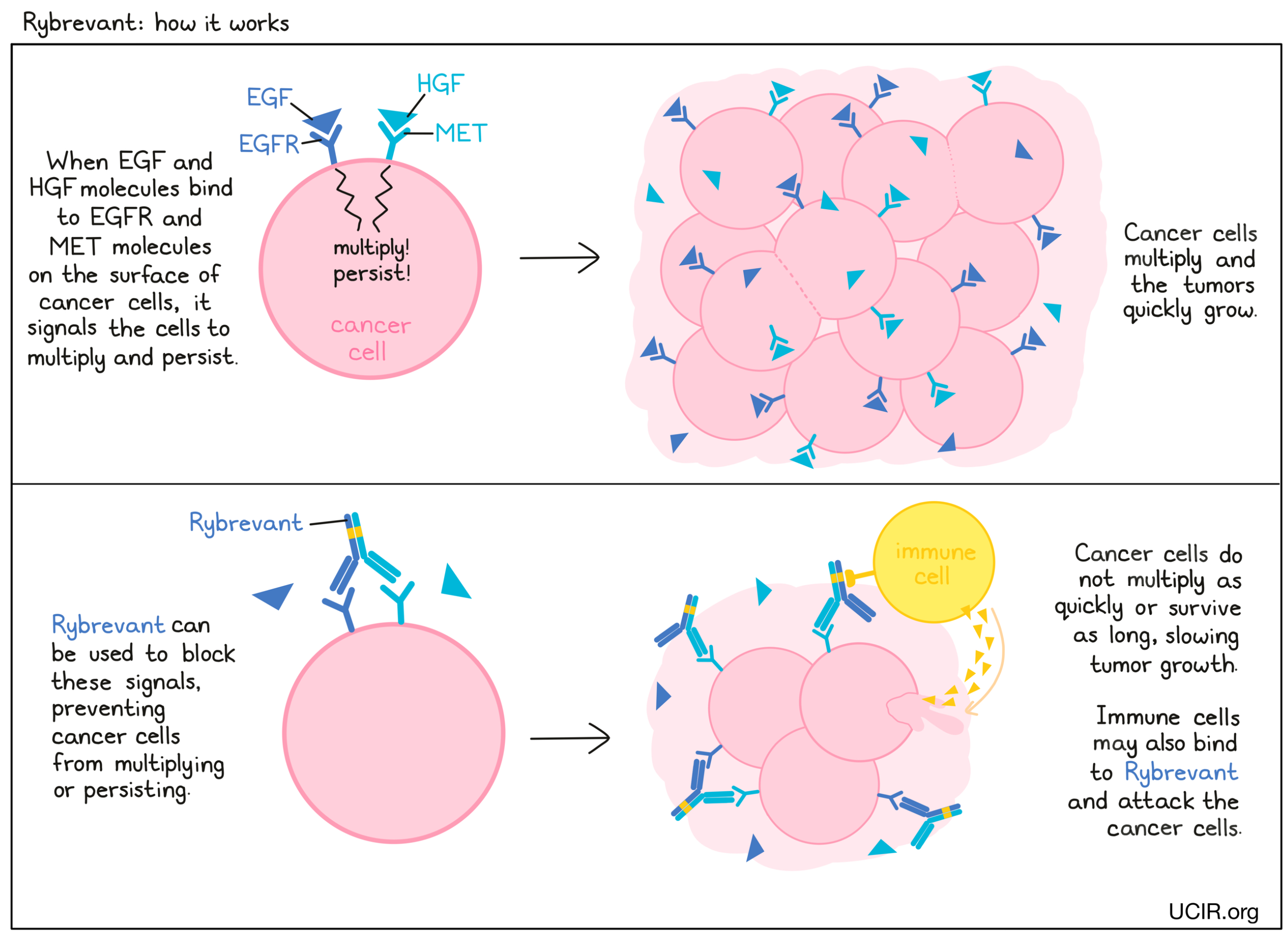
Rybrevant is an antibody that was made in the laboratory and designed to attach to two protein molecules called EGFR (epidermal growth factor receptor) and MET (the hepatocyte growth factor receptor). EGFR and MET are present on the surface of many normal cells (such as cells in the skin, hair follicles, and liver), and are also present in much higher quantities on the surface of many types of cancer cells, including non-small cell lung cancer. Higher-than-normal amounts of EGFR and MET on cancer cells make these cells the main target of Rybrevant. Rybrevant has only been tested and approved for patients with non-small cell lung cancer who carry a particular (mutated) version of EGFR.
Rybrevant and other antibody molecules have an overall “Y” shape. The two tips of the upper arms of the “Y” shape are the parts of the antibody that can very precisely bind to their targets. The stem of Rybrevant’s “Y” shape can attract immune cells or other parts of the immune system.
Rybrevant works to kill cancer cells in at least two ways.
Cancer cell growth inhibition
When certain molecules (such as epidermal growth factor [EGF] and hepatocyte growth factor [HGF]) bind to EGFR and MET on the surface of cells, the cells receive signals to ensure their survival and encourage the cells to grow and multiply. Higher-than-normal amounts of EGFR and MET allow cancer cells to grow and multiply out of control. By binding to these molecules, Rybrevant blocks these signals and prevents the cells from persisting and multiplying.
Antibody-dependent cell-mediated cytotoxicity (ADCC)
When bound to EGFR or MET on the surface of cancer cells, the “stem” of Rybrevant can also attract and bind immune cells (like NK cells). This allows Rybrevant to act as a bridge between the target cell and the immune cell. The immune cell then releases molecules that can kill the cell Rybrevant is bound to.
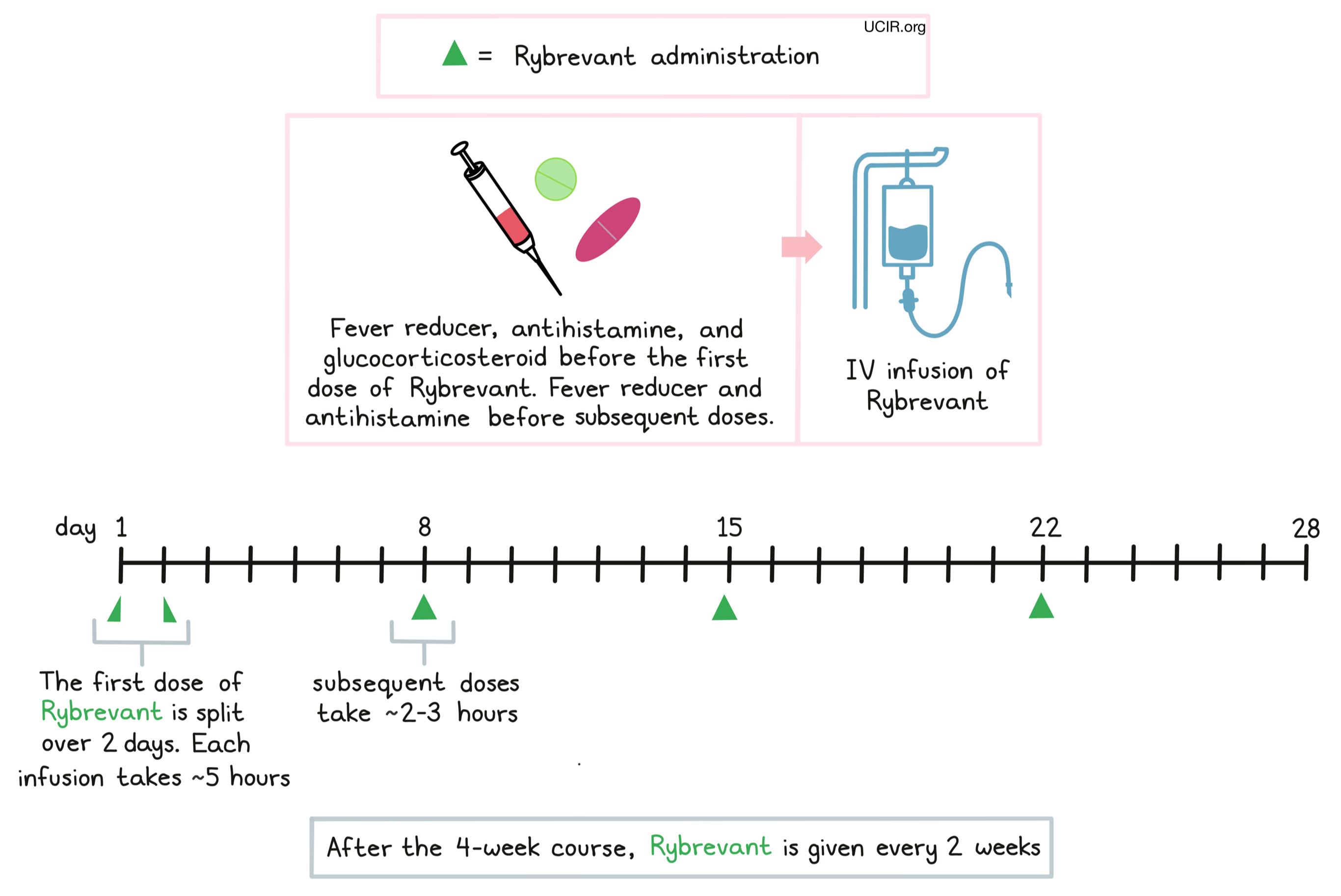
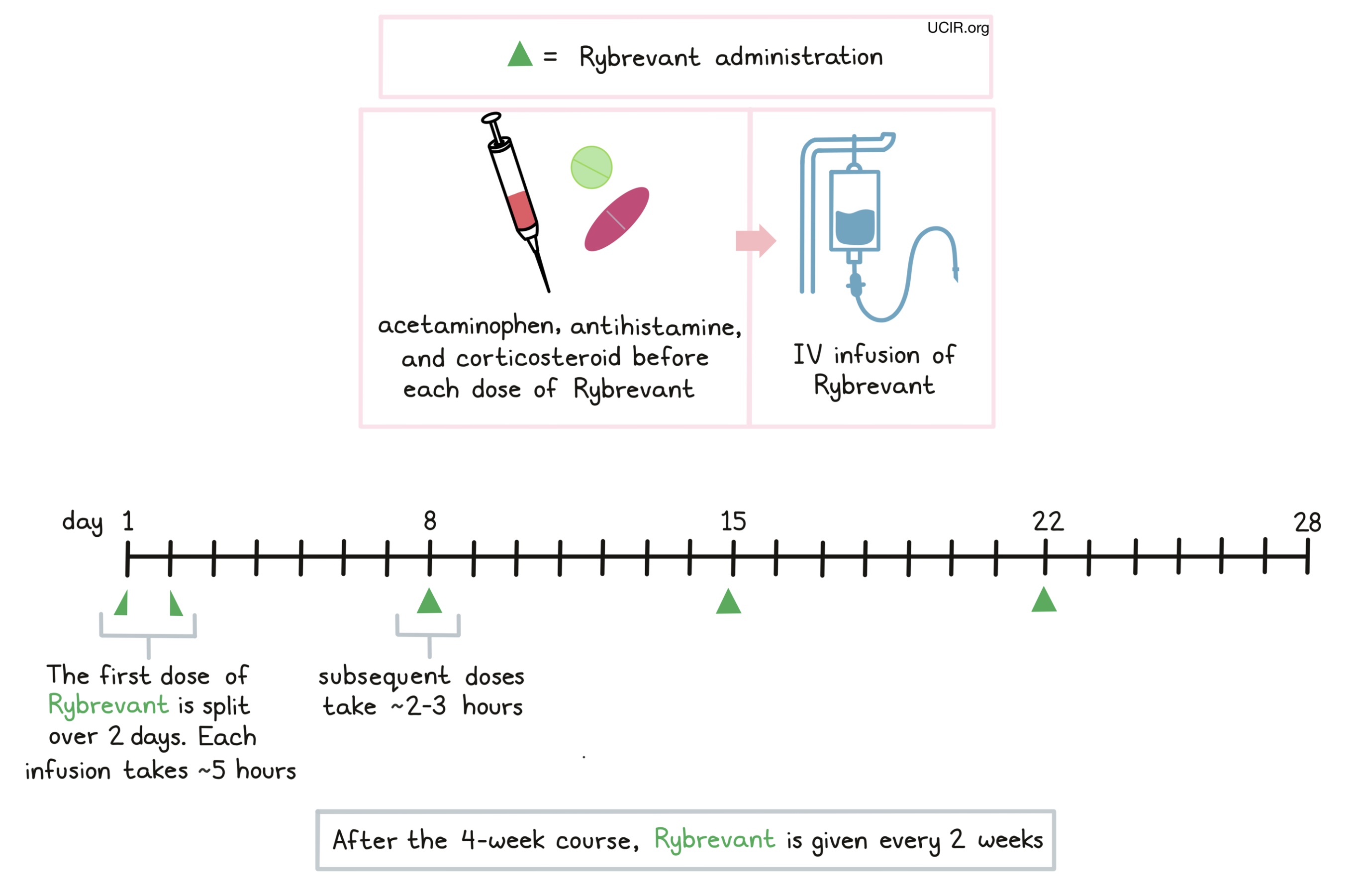
How is this drug given to the patient?
Rybrevant is administered via a tube in the vein (intravenous infusion, or i.v.) once a week, for 4 weeks. The first dose is split across two infusions, administered over 5 hours on both Day 1 and Day 2 of the first week. Subsequent infusions are administered on Day 1 of each week, and take 2-3 hours. After the 4-week course, Rybrevant is administered once every 2 weeks until disease progresses and gets worse, or the side effects become unmanageable.
Prior to each infusion, patients receive acetaminophen, an antihistamine (diphenhydramine) and a corticosteroid (e.g. dexamethasone or methylprednisolone) to reduce the risk of reactions to the infusion. During administration of Rybrevant, patients should be closely monitored for reactions to the infusion. If symptoms of an infusion-related reaction occur, the infusion may be interrupted or slowed, depending on the severity of the reaction.
What are the observed clinical results?
It is important to keep in mind that each patient’s actual outcome is individual and may be different from the results found in the clinical studies. In addition, with immunotherapy, sometimes it takes several months for responses to be observed.
Advanced non-small cell lung cancer
In a clinical trial, 81 patients with EGFR-positive advanced non-small cell lung cancer who had previously received treatment with a platinum-based chemotherapy, were treated with Rybrevant. 40% of patients responded to the treatment, and continued to respond for a median of 11 months. 4% of patients saw their cancer disappear entirely.
What are the potential side effects?
The most common side effects of Rybrevant include coughing, shortness of breath, nausea and vomiting, fatigue, constipation, rashes, swelling (including swelling around the finger or toe nails, mouth, and lips), pain in the muscles, joints, and bones, changes in certain blood tests, and reactions related to the infusion.
Some of the potentially serious side effects related to Rybrevant include infusion-related reactions, severe skin reactions (which could lead to severe infections), and problems with the lungs or eyes.
Infusion-related reactions
Infusion-related reactions are side effects related to receiving an infusion. Most infusion-related reactions typically occur within 24 hours of the first or second infusion. Symptoms of an infusion-related reaction include hives or rash, itchiness, swelling of the tongue, lips, face, or throat, coughing, shortness of breath, wheezing, difficulty breathing, weakness, dizziness, feeling as if your heart is fluttering or racing, and chest pain.
Severe skin reactions
Rybrevant may cause serious skin reactions, including toxic epidermal necrolysis. Toxic epidermal necrolysis is characterized by fever and reddening of the skin. The skin is often painful to the touch, and begins to blister and die, sloughing off. Exposure to sunlight could worsen skin reactions, and patients are advised to limit sun exposure and wear sunscreen and sun-protective clothing during and for up to two months after the last dose of Rybrevant.
Lung problems
Rybrevant may cause inflammation of the lungs (pneumonitis) and scarring of the lungs (interstitial lung disease). Symptoms of pneumonitis include cough, shortness of breath, fatigue, loss of appetite, and weight loss. Symptoms of interstitial lung disease include a dry cough and shortness of breath.
Eye problems
Rybrevant may cause inflammation of the eyes, blurred or worsening vision, dry, itchy, or red eyes.
Patients should report any symptoms to their healthcare provider, who can then initiate actions to limit or reverse the side effects. For a more complete list of possible side effects, see the full prescribing information.
Additional Information
Manufacturer
Janssen Biotech
Approval
FDA and EMA
Links to drug websites
Last updated: January 10, 2023