How is the drug name pronounced?
Belantamab Mafodotin: beh-LAN-tuh-mab MA-foh-DOH-tin
Blenrep: blen-rep
What cancer(s) does this drug treat?
Multiple Myeloma
Blenrep is approved for:
- Patients with multiple myeloma who have received at least four prior treatments for their disease, including an anti-CD38 antibody (e.g., daratumumab (Darzalex) or isatuximab-irfc (Sarclisa)), a proteasome inhibitor, and an immunomodulatory agent, but whose cancer either did not respond to treatment or has since come back.
Limitations of Use
Blenrep REMS program: Due to the risk of potentially serious side effects, Blenrep is currently only available through a Risk Evaluation and Mitigation Strategy (REMS) program. Hospitals or clinics that dispense Blenrep require certification for the program, ensuring proper monitoring and management of side effects associated with the drug.
Age: The safety and efficacy of Blenrep has not been established in patients under 18 years of age.
Fertility/Pregnancy/breastfeeding: Blenrep may impair fertility in women and in men. In pregnant women, Blenrep may cause harm to the fetus and is not recommended for use during pregnancy. Women are advised to use contraception during treatment with Blenrep and for at least 4 months after the last dose of Blenrep. The risks associated with Blenrep during breastfeeding are not known and cannot be ruled out. Due to the potential for serious adverse reactions in the breastfed child, women are advised not to breastfeed during treatment with Blenrep and for up to 3 months after the last dose of Blenrep. Men are advised to use contraception during treatment with Blenrep and for at least 6 months after the last dose of Blenrep.
What type of immunotherapy is this?
- Antibody–drug conjugate
How does this drug work?
Target:
- BCMA molecule on B cells
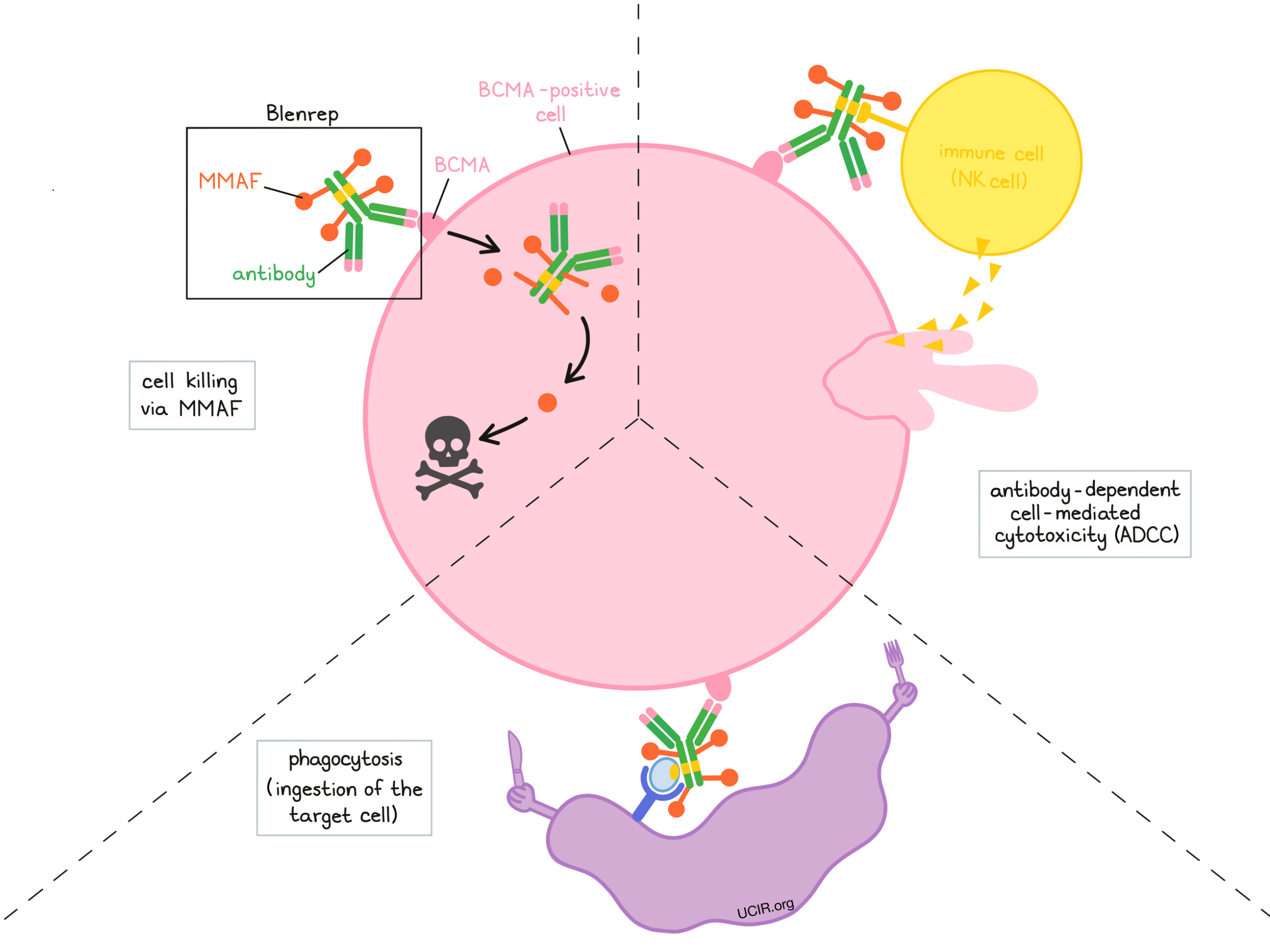
Blenrep is a medicine that consists of two parts that are connected:
- an antibody that attaches to a molecule called B-cell maturation antigen (BCMA) on the surface of multiple myeloma cells and some healthy B cells, and
- a chemotherapy drug called monomethyl auristatin F (MMAF)
Blenrep and other antibody molecules have an overall “Y” shape. The two tips of the upper arms of the “Y” shape are the parts of the antibody that can very precisely bind to their targets, in this case, BCMA. The stem of Blenrep’s “Y” shape has the chemotherapy drug MMAF connected to it, but also has binding sites for immune cells or other parts of the immune system.
At least three different mechanisms are thought to be responsible for the elimination of BCMA-positive multiple myeloma cells by Blenrep. Blenrep may work alone or may be helped by the immune system to kill multiple myeloma cells.
Cell killing via MMAF
When Blenrep binds to BCMA, it can enter the cell it is bound to. Inside the cell, the chemotherapy part of Blenrep is released and becomes activated. MMAF prevents the cancer cells from multiplying and leads to their death. By attaching to BCMA, Blenrep is designed to minimize harm to normal, healthy cells, and to bring the chemotherapy directly to the cancer cells.
Antibody-dependent cell-mediated cytotoxicity (ADCC)
When bound to BCMA on the surface of multiple myeloma cells, the “stem” of Blenrep can also attract and bind immune cells (like NK cells). This allows Blenrep to act as a bridge between the target cell and the immune cell. The immune cell then releases molecules that can kill the cell Blenrep is bound to.
Phagocytosis
When Blenrep is bound to BCMA on the surface of multiple myeloma cells, it can attract immune cells called phagocytes that have the ability to ingest (“eat”) cells that have been coated with Blenrep or with parts of the complement system.
How is this drug given to the patient?
Blenrep is administered through a tube in the vein (intravenous infusion, or i.v.) over 30 minutes once every 3 weeks.
Due to the risk of damage to the eyes, patients should have an eye examination before starting treatment with Blenrep, prior to each dose of Blenrep, and promptly if eye problems arise. Treatment with Blenrep may be withheld or permanently stopped based on the severity and potential improvement of the eye problems. Prior to starting Blenrep treatment, patients also get blood work done.
Patients are advised to use preservative-free lubricant eye drops at least 4 times a day and avoid the use of contact lenses during treatment with Blenrep.
What are the observed clinical results?
It is important to keep in mind that each patient’s actual outcome is individual and may be different from the results found in the clinical studies described below. In addition, with immunotherapy, sometimes it takes several months for responses to be observed.
Multiple myeloma
In a clinical trial, 97 patients with multiple myeloma who had received with at least three prior therapies for disease (including an anti-CD38 monoclonal antibody), but whose cancer either did not respond to treatment (including treatment with a proteasome inhibitor and an immunomodulatory agent) or had since come back, were treated with Blenrep. At a median follow-up of 6 months, 31% of patients responded to the treatment, with 3% seeing their cancer disappear entirely. The median time to first response was 1 month, and 73% of responding patients continued to respond to the treatment for at least 6 months.
What are the potential side effects?
The most common side effects with Blenrep include eye problems, nausea, fatigue, fever, reactions related to Blenrep infusion, diarrhea, constipation, back pain, joint pain, upper respiratory tract infections, unusual bleeding, low platelet count, low white blood cell count, and other abnormal blood test results.
Eye problems
Blenrep can damage the corneal epithelium, the clear layer of tissue covering the pupil and iris. This can result in dry eyes, corneal ulcers, blurry vision, worsening vision, and vision loss. Eye exams will be conducted prior to and during treatment in order to assess whether any corneal damage has taken place. If problems arise, the administration of Blenrep is interrupted until the symptoms improve, or is permanently stopped.
For a more complete list of possible side effects, see the full prescribing information.
Additional Information
Manufacturer
GlaxoSmithKline (GSK)
Approval
FDA and EMA
Link to drug website
Other references
Last updated March 8, 2022


