How is this drug name pronounced?
Glofitamab: gloh-FIH-tah-mab
Columvi: koh-LOOM-vee
What cancer(s) does this drug treat?
Diffuse large B cell lymphoma (DLBCL) and large B cell lymphoma (LBCL)
Columvi is approved for:
- Patients with diffuse large B-cell lymphoma (DLBCL), not otherwise specified, or large B cell lymphoma (LBCL) arising from follicular lymphoma (FL), who have received at least two prior treatments for their disease, but whose cancer either did not respond to treatment or has since come back.
Limitations of use
Limitations: Columvi should only be administered in a treatment center and by adequately trained healthcare professionals who can ensure proper monitoring and immediate management of side effects associated with the drug.
Age: The safety and efficacy of Columvi in patients under 18 years of age have not been established.
Pregnancy/Breastfeeding: Columvi is not recommended for use during pregnancy due to potential for harm to the fetus. Patients are advised to use contraception during treatment with Columvi and for one month after the last dose of Columvi. The risks associated with Columvi during breastfeeding are not known and cannot be ruled out. Due to the potential for adverse reactions in the breastfed child, patients are advised not to breastfeed during treatment with Columvi and for one month after the last dose of Columvi.
Interactions with other drugs: Dose adjustments may become necessary for patients treated with certain CYP substrates at the start of treatment with Columvi.
What type of immunotherapy is this?
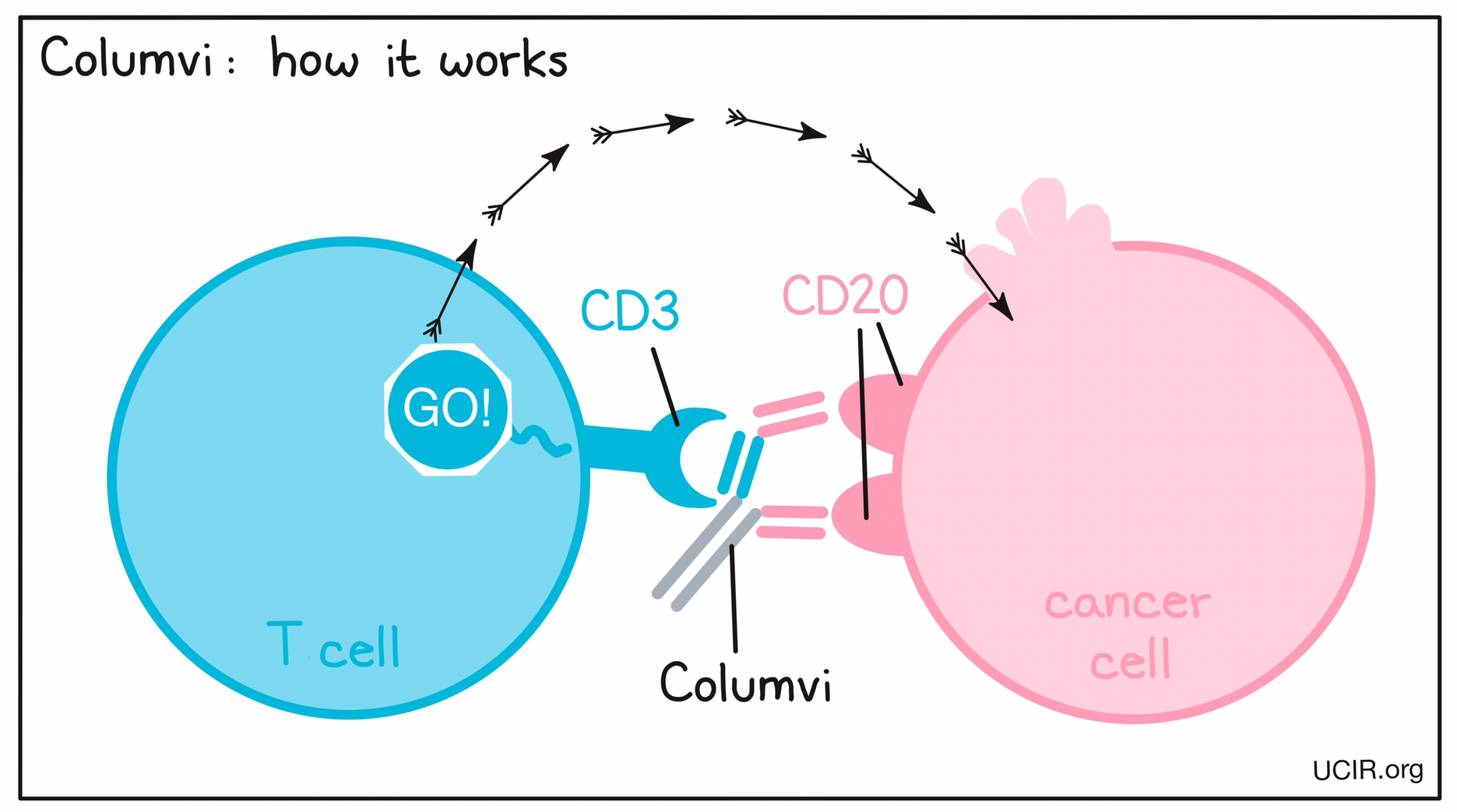
How does this drug work?
Targets:
- CD20 on B cells
- CD3 receptor on T cells
Columvi is a “bispecific antibody”. Antibodies are molecules that can bind to a particular target molecule. Bispecific antibodies are artificially made in the laboratory and can bind to two different targets at the same time. Columvi binds to:
- a molecule called CD20 on the surface of lymphoma cells and some healthy B cells. CD20 is present at much higher quantities on the surface of cancer cells than normal healthy cells.
- a molecule called CD3 on the surface of T cells – the primary immune cells involved in killing cancer cells. CD3 is part of the T cell receptor, which is critical to the function of the T cell and is involved in stimulating the T cell to become active.
Because Columvi can bind to one molecule on lymphoma cells and another on T cells at the same time, it acts as a bridge and keeps the T cell in close contact with the cancer cell. By binding CD3 on the T cell, Columvi also stimulates the T cell to become activated and kill the cell it is bound to. Bispecific antibodies that direct T cells to kill cancer cells by binding to both cells at the same time are known as bispecific T cell engagers (BiTEs).
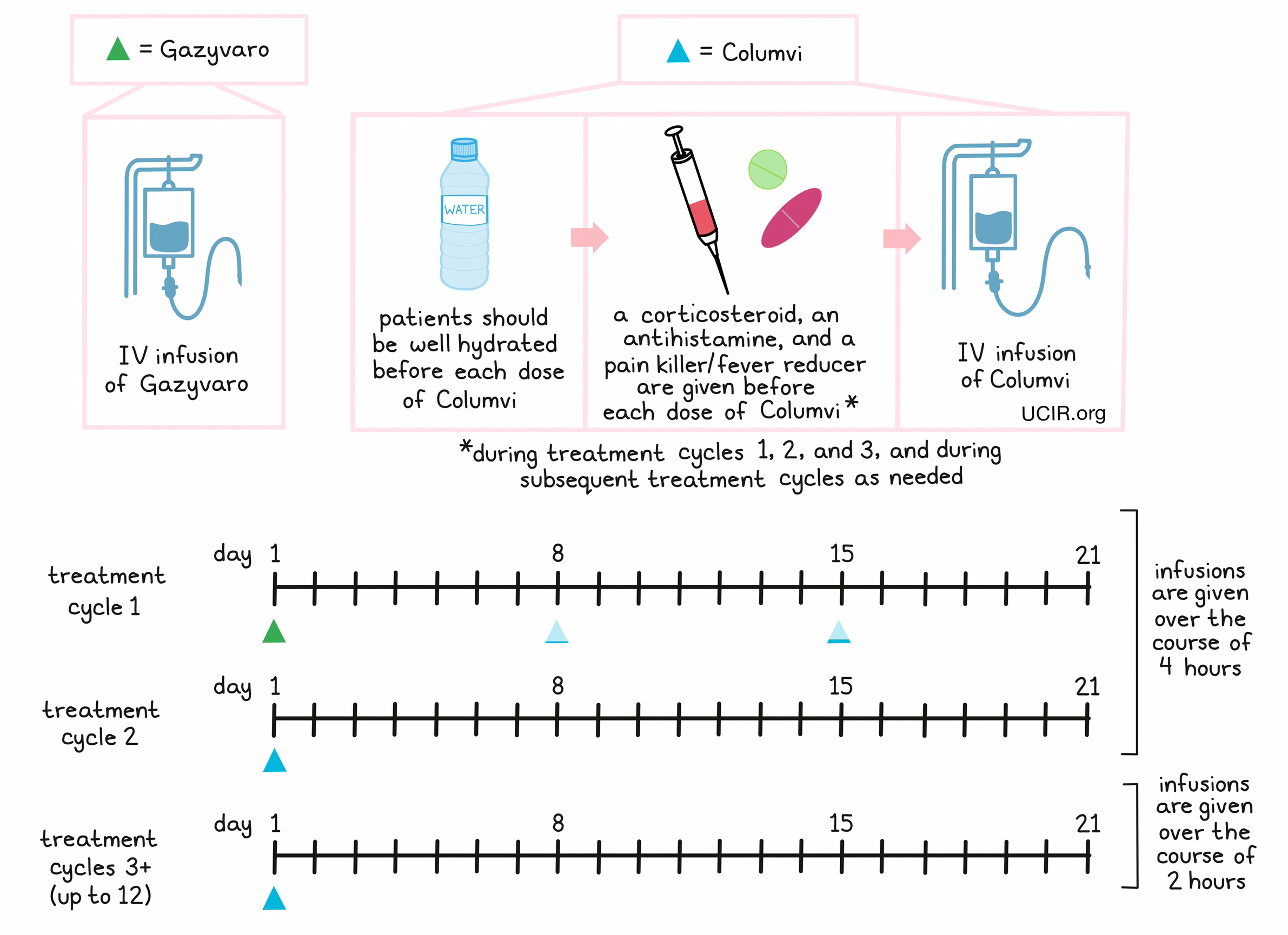
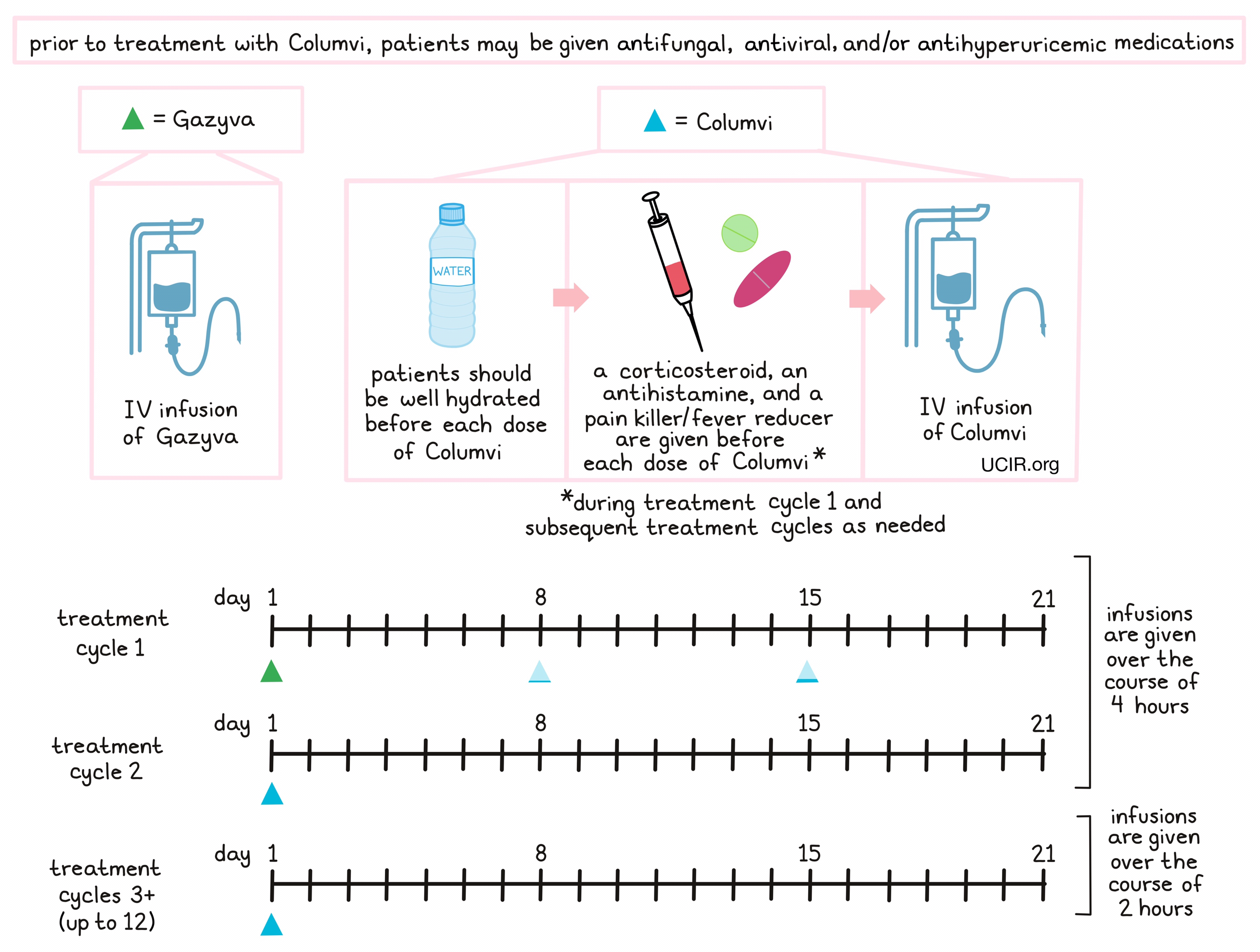
How is the drug given to the patient?
Prior to starting treatment with Columvi, patients may receive antifungal medication to prevent a certain form of pneumonia, an antiviral medication to prevent herpes zoster (shingles) reactivation and/or cytomegalovirus infection, and/or antihyperuricemic medications to limit tumor lysis syndrome. Patients should be well hydrated before they receive each dose of Columvi. About 1 hour to 30 minutes prior to receiving each dose of Columvi in treatment cycle 1 and, if needed, in subsequent treatment cycles, patients receive several medications to further reduce the risk of cytokine release syndrome:
- A corticosteroid (dexamethasone)
- An antihistamine (diphenhydramine)
- A painkiller and fever reducer (acetaminophen)
Columvi is administered through a tube in the vein (intravenous [IV]) infusion over the course of 4 hours during the first 2 treatment cycles, or 2 hours during subsequent treatment cycles. During the first treatment cycle (a 21-day period), obinutuzumab (Gazyva) is administered on day 1 by IV infusion to limit cytokine release syndrome (a common and potentially severe side effect of Columvi treatment). A low dose of Columvi is administered on day 8, and a second, slightly higher dose of Columvi is given on day 15. For every subsequent treatment cycle, a full dose of Columvi is given on day 1. Patients may receive up to 12 treatment cycles.
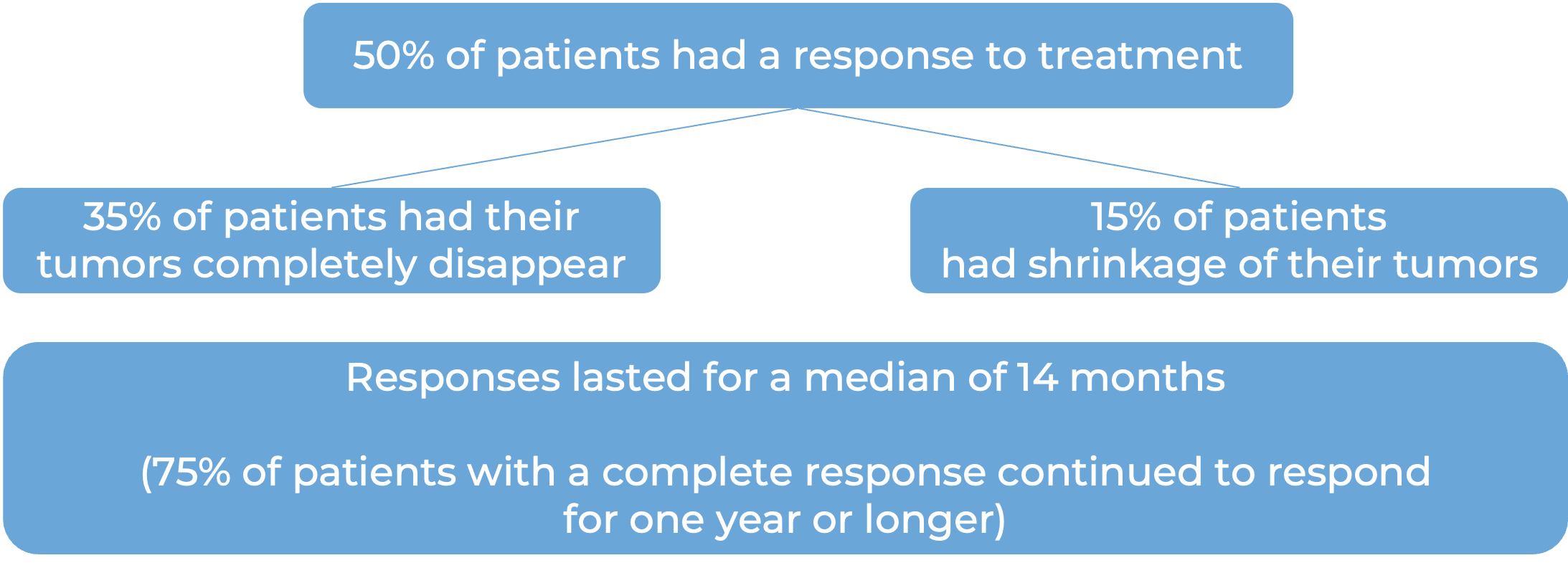
What are the observed clinical results?
It is important to keep in mind that each patient’s outcome is individual and may be different from the results found in the clinical studies. In addition, immunotherapy can sometimes take several months to yield an observable treatment response.
Diffuse large B cell lymphoma or large B cell lymphoma
In a clinical study, 132 patients with diffuse large B-cell lymphoma (DLBCL), not otherwise specified, or large B cell lymphoma arising from follicular lymphoma who had received at least two prior treatments for their disease, but whose cancer either did not respond to treatment or had since come back, were treated with Columvi.
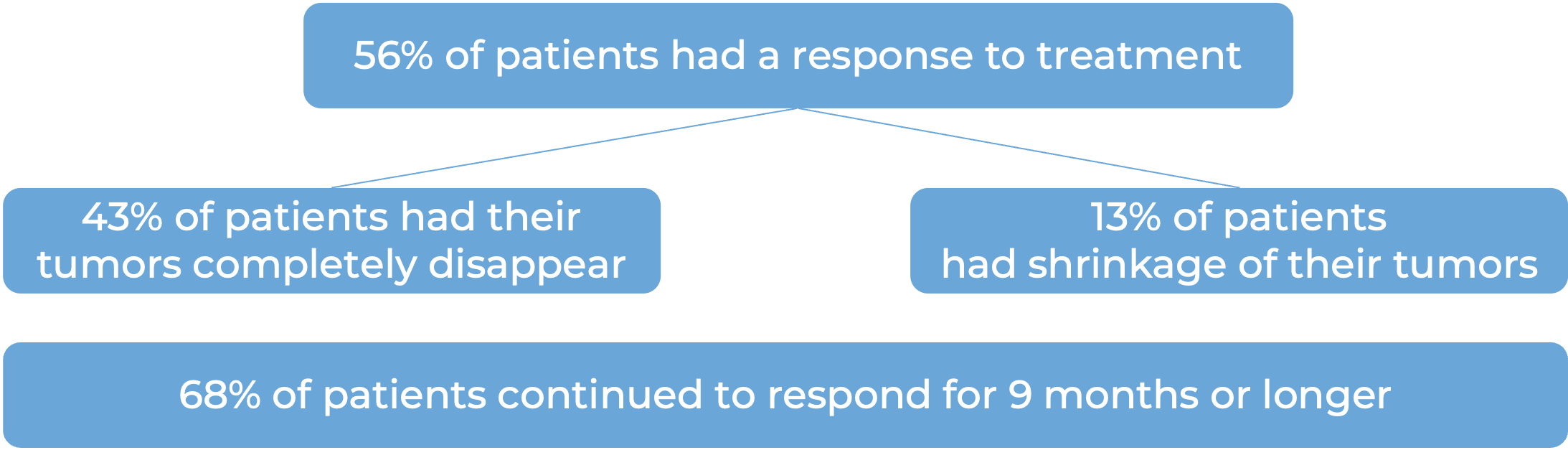
(For a definition of median click HERE.)
What are the potential side effects?
Columvi targets CD20, which, while present on lymphoma cells, is also present on some normal B cells. As a result, Columvi can also kill normal B cells, increasing the risk of serious infections. Other common side effects of Columvi include cytokine release syndrome, pain in the muscles, joints, and bones, fatigue, rash, low white blood cell count, and other abnormal blood test results. Some side effects, such as cytokine release syndrome, immune effector cell-associated neurotoxicity syndrome, tumor flare, infections, or abnormal blood counts, can become serious or life-threatening. Patients and caregivers receive careful instructions to monitor for signs and symptoms of side effects. These conditions are managed by the healthcare provider.
Cytokine release syndrome (CRS)
CRS is caused by a widespread release of molecules called cytokines, which can cause inflammation and can affect the function of various organs. Cytokines may be released by the T cells to which Columvi binds, or by other immune cells in the patient’s body. Signs and symptoms of CRS include fever, low blood pressure, hypoxia (low oxygen levels), fast heart rate, and chills. CRS typically occurs within 24 hours after the most recent dose of Columvi in treatment cycle 1. A healthcare provider should be immediately notified if symptoms occur.
Immune effector cell-associated neurotoxicity syndrome (ICANS)
Some of the cytokines released during CRS can result in disruption of the blood–brain barrier, leading to the development of neurological toxicities. Symptoms include headache, dizziness, vertigo, shaking, lethargy, seizures, confusion, and difficulty with speech or handwriting. A healthcare provider should be notified immediately if symptoms occur.
Tumor flare (transient growth of the tumor or worsening of tumor-related problems) Columvi can cause swelling or pain at the sites of lymphoma lesions. Tumor flare events typically occur within 2 days after the first dose of Columvi and last for a median of 3 to 4 days.
For a more complete list of possible side effects, see the full prescribing information.
Additional information
Manufacturer
Genentech
Approval
FDA and EMA
Links to drug websites
Last updated: August 8, 2025