How is the drug name pronounced?
Perjeta: PER-jet-ah
Pertuzumab: per-TOO-zoo-mab
What cancer(s) does this drug treat?
Early breast cancer
Perjeta is approved for:
- Patients with breast cancer that has spread within the breast or to the lymph nodes under the arms, but not to other parts of the body; is greater than 2 cm in diameter or inflammatory; and tests positive for high amounts of the HER2 molecule. In such cases, Perjeta is used in combination with trastuzumab (e.g., Herceptin) and chemotherapy before surgical removal of all known disease.
- Patients with breast cancer that tests positive for high amounts of the HER2 molecule, and who are at high risk for their cancer coming back after surgical removal of all known disease. In such cases, Perjeta is used in combination with trastuzumab (e.g., Herceptin) and chemotherapy.
Advanced breast cancer
Perjeta is approved for:
- Patients with breast cancer that has spread to other parts of the body and tests positive for high amounts of the HER2 molecule, and who have not received prior treatments that target the HER2 molecule, or chemotherapy for their advanced disease. In such cases, Perjeta is used in combination with trastuzumab (e.g., Herceptin) and docetaxel chemotherapy.
Limitations of Use
Age: The safety and efficacy of Perjeta in patients under 18 years of age have not been established.
Pregnancy/Breastfeeding: Treatment with Perjeta can cause serious side effects and death to a fetus, and is not recommended for use during pregnancy. Women are advised to use contraception during treatment with Perjeta and for at least 7 months after the last dose of Perjeta. Women who receive Perjeta while pregnant or within 7 months before conception should be monitored for any signs of an insufficient volume of the amniotic fluid. The risks associated with Perjeta during breastfeeding are not known and cannot be ruled out. Due to the potential for adverse reactions in the breastfed child, women are advised not to breastfeed during treatment with Perjeta and within 7 months after the last dose of Perjeta.
What type of immunotherapy is this?
- Cell growth inhibitor
How does this drug work?
- Target: Human epidermal growth factor receptor 2 (HER2)
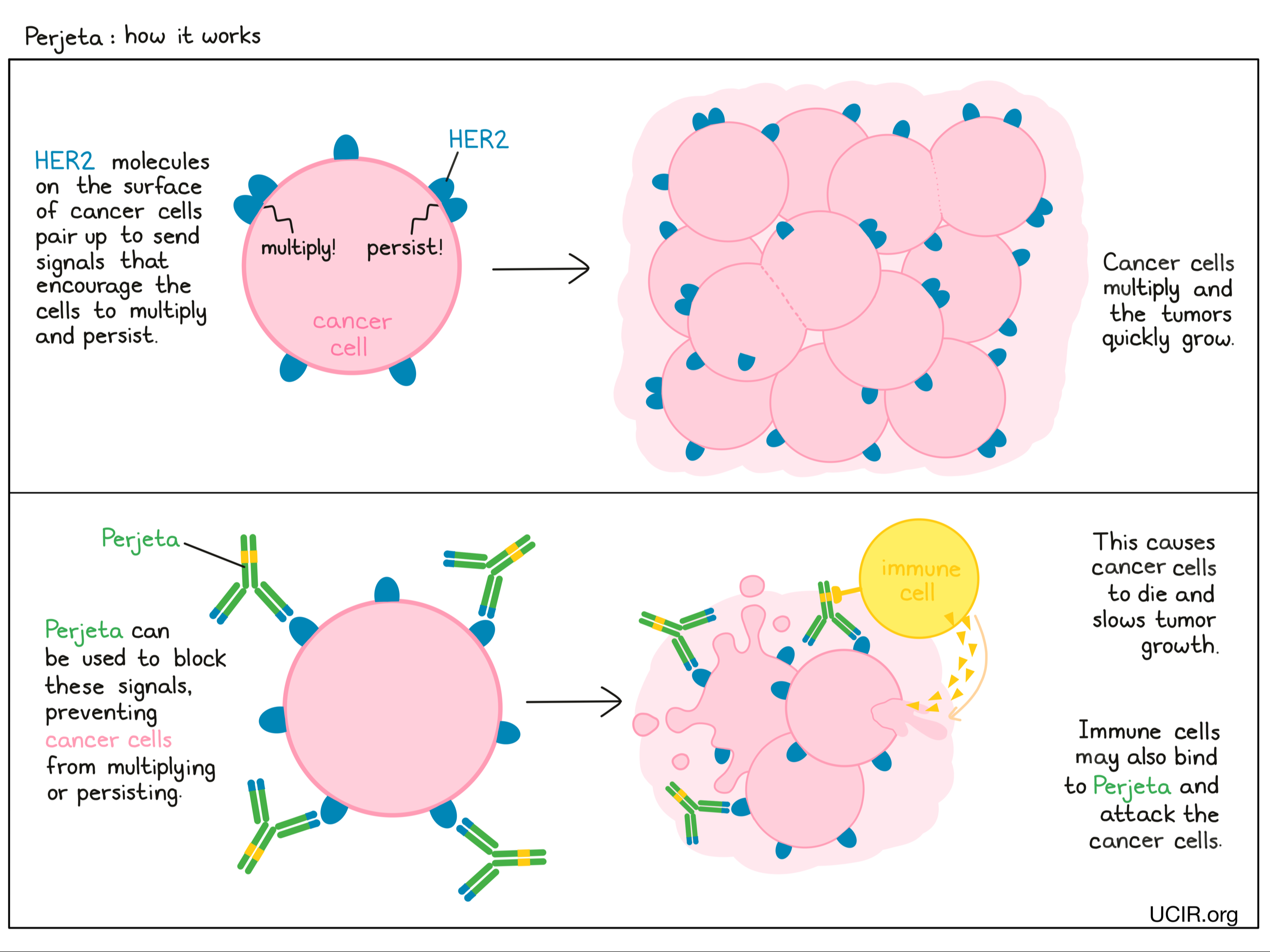
Perjeta is an antibody that was made in the laboratory and was designed to attach to a protein molecule called HER2. HER2 is present on the surface of some normal cells in the body, and it is also present in much higher quantities on the surface of breast cancer cells. Higher-than-normal amounts of HER2 on cancer cells make these cells the main targets of Perjeta.
Perjeta and other antibodies have an overall “Y” shape. The two tips of the upper arms of the “Y” shape are the parts of the antibody that can very precisely bind to their targets. For Perjeta, the tips of the upper arms bind to HER2. The stem of Perjeta’s “Y” shape can attract immune cells or other parts of the immune system.
Perjeta works to kill cancer cells in at least two ways.
Cancer cell growth inhibition and cancer cell death
When HER2 on the surface of cells binds to itself or to other HER2-related proteins, it sends signals into the cell. These signals cause the cell to multiply and persist. Higher-than-normal amounts of HER2 allow cancer cells to grow and multiply out of control. Binding of Perjeta to HER2 blocks HER2 from pairing up and sending these signals into the cancer cells, preventing the cells from persisting and multiplying, and forcing HER2-positive breast cancer cells to undergo “programmed cell death”.
Antibody-dependent cell-mediated cytotoxicity (ADCC)
When bound to HER2 on the surface of breast cancer cells, the stem of Perjeta can also attract and bind immune cells. This allows Perjeta to act as a bridge between the target cell and an immune cell (such as a natural killer (NK) cell). The immune cell then releases molecules that can kill the cell that Perjeta is bound to.
Perjeta and trastuzumab (e.g., Herceptin) bind the HER2 molecule on different sides. When administered together, both drugs can achieve a greater benefit than Perjeta or trastuzumab alone.
How is this drug given to the patient?
Before starting treatment with Perjeta, a small tumor sample is collected from patients and tested to determine if the tumor tests positive for high amounts of the HER2 molecule. Patients also undergo a thorough assessment of the condition of their heart before starting treatment with Perjeta, during treatment, and upon completion of treatment.
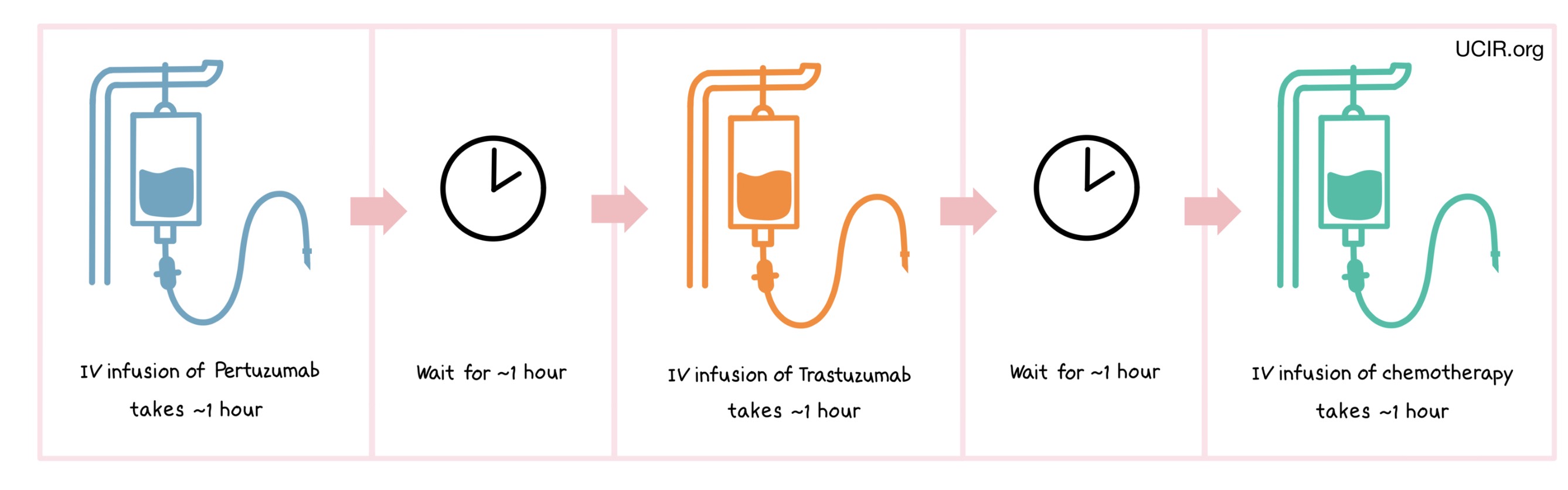
Perjeta is administered every 3 weeks through a tube in the vein (intravenous infusion or i.v.) and does not require a hospital stay. Perjeta is administered in combination with other drugs. Usually Perjeta and trastuzumab (e.g., Herceptin) are given in any order before infusion of the chemotherapy. Each drug is given one at a time as an infusion over the course of 30-90 minutes each, with a wait period of 30-60 minutes between infusions. When given as a treatment for early breast cancer, patients receive 3 to 6 doses of Perjeta at the 3-week interval before complete surgical removal of all known disease. Patients then continue treatment after surgery to complete a full year of therapy. Treatment may be discontinued before this point if the disease progresses and gets worse, or if the side effects become unmanageable.
What are the observed clinical results?
For:
Early breast cancer
Advanced breast cancer
It is important to keep in mind that each patient’s prognosis is individual and may be different from the results found in the clinical studies. In addition, with immunotherapy, it sometimes takes several months for responses to be observed.
Early breast cancer
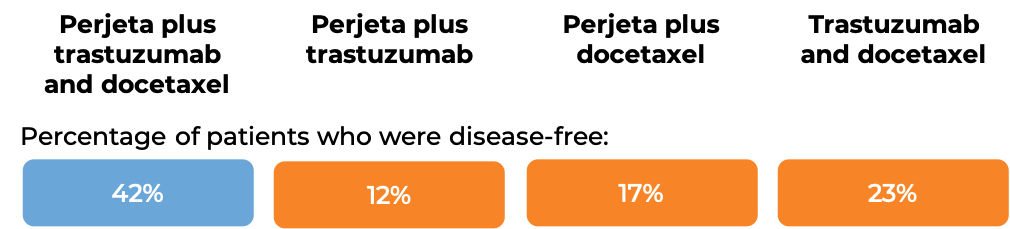
In a clinical trial, 417 patients with breast cancer that had spread within the breast or to the lymph nodes under the arms, but not to other parts of the body; was inflammatory; and tested positive for high amounts of the HER2 molecule, received one of four treatments prior to surgery:
- Perjeta in combination with trastuzumab (e.g., Herceptin) and docetaxel,
- Perjeta in combination with trastuzumab,
- Perjeta in combination with docetaxel, OR
- trastuzumab and docetaxel only.
Following surgery, all patients received 3 treatment cycles of 5-fluorouracil, epirubicin, and cyclophosphamide, and after that, trastuzumab every 3 weeks to complete 1 year of therapy. (Patients who were treated with Perjeta plus trastuzumab before surgery received 4 treatment cycles of docetaxel prior to starting the 3 treatment cycles of 5-fluorouracil, epirubicin, and cyclophosphamide, and trastuzumab every 3 weeks to complete 1 year of therapy).
In another clinical trial, 401 patient with breast cancer that had spread within the breast or to the lymph nodes under the arms, but not to other parts of the body; was inflammatory; tested positive for high amounts of the HER2 molecule; and could be removed by surgery, were treated with either:
- Treatment 1: four treatment cycles of doxorubicin and cyclophosphamide followed by four treatment cycles of Perjeta in combination with trastuzumab and weekly paclitaxel for 12 weeks prior to surgery, and Perjeta plus trastuzumab following surgery to complete 1 year of therapy, OR
- Treatment 2: four treatment cycles of 5-fluorouracil, epirubicin and cyclophosphamide followed by 4 treatment cycles of Perjeta in combination with trastuzumab and docetaxel prior to surgery, and Perjeta plus trastuzumab following surgery to complete 1 year of therapy
At a median follow-up of 15 months:

Advanced breast cancer
In a clinical trial, 808 patients with breast cancer that had spread to other parts of the body and tested positive for high amounts of the HER2 molecule were either treated with pertuzumab, trastuzumab (e.g., Herceptin), and docetaxel, or with a placebo, trastuzumab, and docetaxel.
At a median follow-up of 50 months:

What are the potential side effects?
The most common side effects of Perjeta in combination with trastuzumab and chemotherapy include hair loss, rash, nausea, diarrhea, fatigue, nerve damage (tingling, numbness, or pain in hands and feet), low red blood cell count, and low white blood cell count.
Some side effects, such as heart problems, reactions related to the infusion, and hypersensitivity reactions can become serious or life-threatening. Patients and caregivers receive careful instructions to monitor for signs and symptoms related to these conditions. These conditions are managed by the healthcare provider.
Heart problems
Perjeta can cause heart problems, including high blood pressure, uneven heartbeat, dysfunction of the heart’s left ventricle, heart disease, heart failure, and heart attack. Prior to the initiation of treatment with Perjeta, patients undergo a thorough assessment of the condition of their heart. Patients also have their hearts monitored regularly during treatment with Perjeta, and have their treatment discontinued if severe heart issues occur.
Infusion-related reactions
Infusion-related reactions are negative responses to receiving an infusion. Most infusion-related reactions typically occur within 24 hours of the first or second infusion. Symptoms of an infusion-related reaction include hives (itchy red welts) or rash, itchiness (pruritus), and swelling of the tongue, lips, face, or throat. Coughing, shortness of breath, wheezing, difficulty breathing, weakness, dizziness, and faintness can also occur. Palpitations (feeling as if your heart is fluttering or racing) and chest pain can also be symptoms of a reaction.
Hypersensitivity reactions
Hypersensitivity reactions are allergic reactions caused by the body’s immune system responding to Perjeta as if it were a threat. Symptoms of a hypersensitivity reaction may include rashes, itching, wheezing, swelling of the face, shortness of breath, swelling of the airways, and anaphylactic shock.
Patients should report any symptoms to their healthcare provider, who can then initiate actions to limit or reverse the side effects. For a more complete list of possible side effects, see the full prescribing information.
Additional Information
Manufacturer
Genentech/Roche
Approval
FDA and EMA
Links to drug website
Other references
- Pertuzumab, Trastuzumab, and Docetaxel in HER2-Positive Metastatic Breast Cancer. Swain SM et al. New England Journal of Medicine (2015)
- Pertuzumab, trastuzumab, and standard anthracycline- and taxane-based chemotherapy for the neoadjuvant treatment of patients with HER2-positive localized breast cancer (BERENICE): a phase II, open-label, multicenter, multinational cardiac safety study. Swain SM et al. Annals of Oncology (2018)
Last updated on August 8, 2025


