How is this drug name pronounced?
Tremelimumab: treh-meh-LIM-yoo-mab
Imjudo: im-JOO-doh
What cancer(s) does this drug treat?
Advanced liver cancer
Imjudo is approved for:
- Patients with hepatocellular carcinoma (liver cancer) that cannot be removed by surgery. In such cases, patients are treated with Imjudo in combination with durvalumab (Imfinzi).
Advanced non-small cell lung cancer
Imjudo is approved for:
- Patients with non-small cell lung cancer that has spread to other parts of the body and does not have an abnormal EGFR or ALK gene. In such cases, Imjudo is used in combination with platinum-based chemotherapy and durvalumab (Imfinzi).
Limitations of use:
Age: The safety and efficacy of Imjudo in patients under 18 years of age have not been established.
Pregnancy/Breastfeeding: Imjudo can cause harm to a fetus, and is not recommended for use during pregnancy. Patients who are able to become pregnant should use effective birth control during treatment and for at least 3 months after the last dose of Imjudo. The risks associated with Imjudo during breastfeeding are not known and cannot be ruled out; patients are advised not to breastfeed during treatment and for 3 months after the last dose of Imjudo. Patients should also refer to the prescribing info of drugs administered in combination with Imjudo.
What type of immunotherapy is this?
- CTLA-4 blockade
How does this drug work?
- Target: CTLA-4
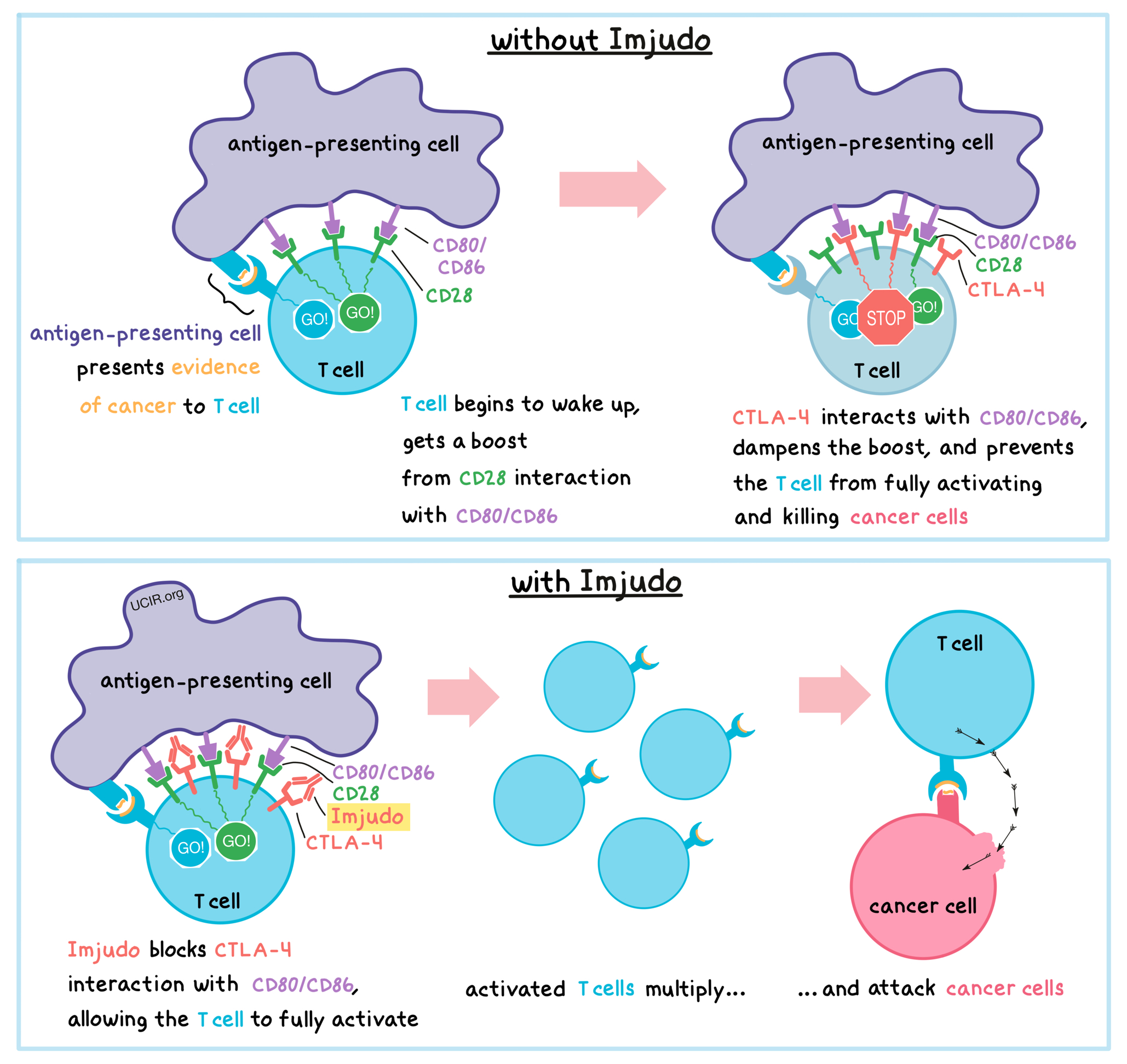
Imjudo is an antibody that attaches to a molecule called CTLA-4, which is present on the surface of T cells – the primary immune cells involved in the killing of cancer cells.
Before T cells can kill cancer cells, they first need to be “woken up”, or activated, and then they have to multiply to launch a potent attack. To help activate T cells, vigilant antigen-presenting cells patrol the body, pick up evidence of cancer, and travel to the lymph nodes to present this evidence to the T cells. If a particular T cell recognizes the evidence of cancer, it begins to wake up. However, to be fully activated for the cancer battle, T cells need a “caffeine” boost. This boost occurs when an activating molecule called CD28 on the surface of T cells interacts with molecules called CD80 or CD86 on the surface of antigen-presenting cells. However, shortly after the boost occurs, CTLA-4 shows up on the surface of T cells and competes with CD28 for the attention of CD80 or CD86, keeping the T cells from getting too much of the “caffeine” boost. Thus, CTLA-4 acts as a brake that keeps the T cells from creating an immune reaction that gets out of control, which could damage healthy tissues. Unfortunately, in cancer, the interaction between CTLA-4 and CD80/CD86 can prevent T cells from becoming activated enough to kill the cancer cells.
Imjudo binds to the CTLA-4 molecules on T cells in such a way that blocks the interaction between CTLA-4 and CD80/CD86 and allows the CD28 molecules on T cells to keep interacting with CD80/CD86 to fully activate the T cells, which is needed to attack cancer. Once the T cells are fully activated, they multiply to create a large army of potent T cells that can hunt down cancer cells throughout the body and kill them wherever they are found.
There may be other ways in which Imjudo helps destroy the cancer, and they are currently under investigation.
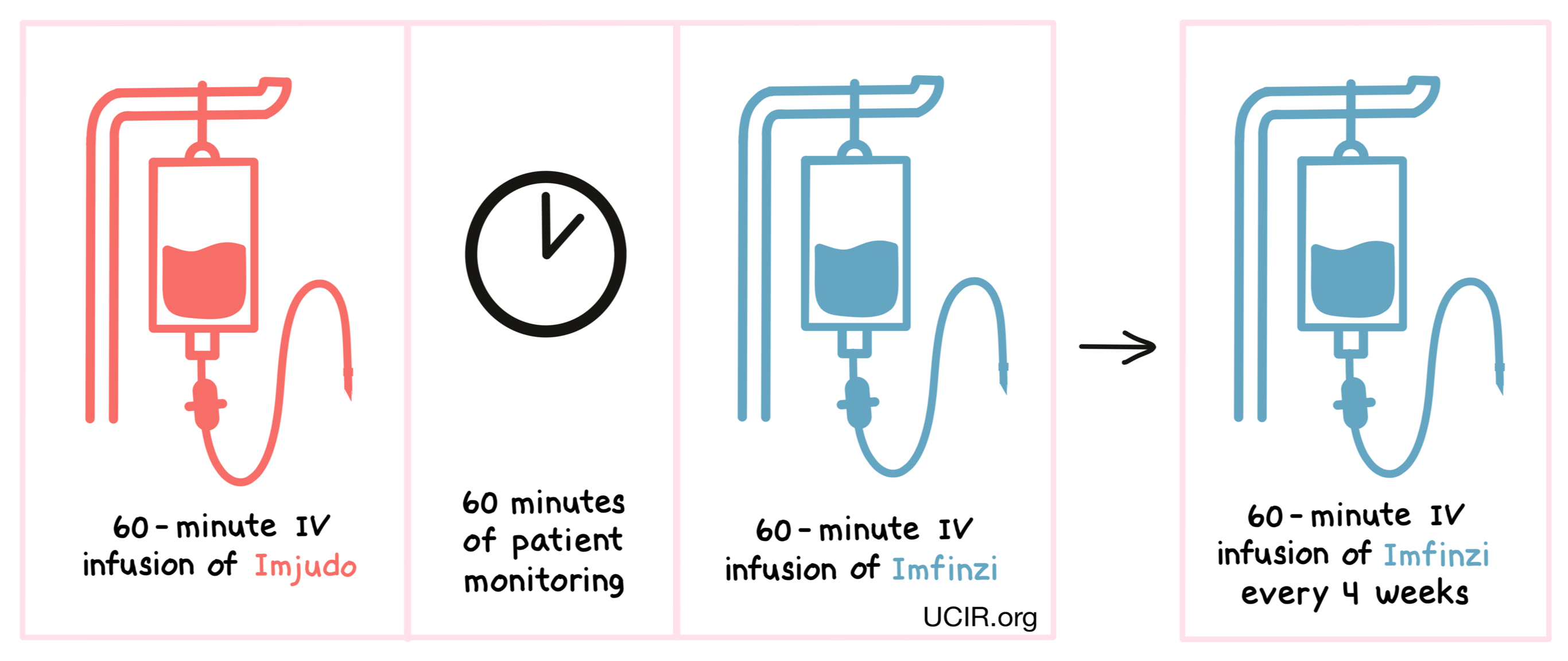
How is this drug given to the patient?
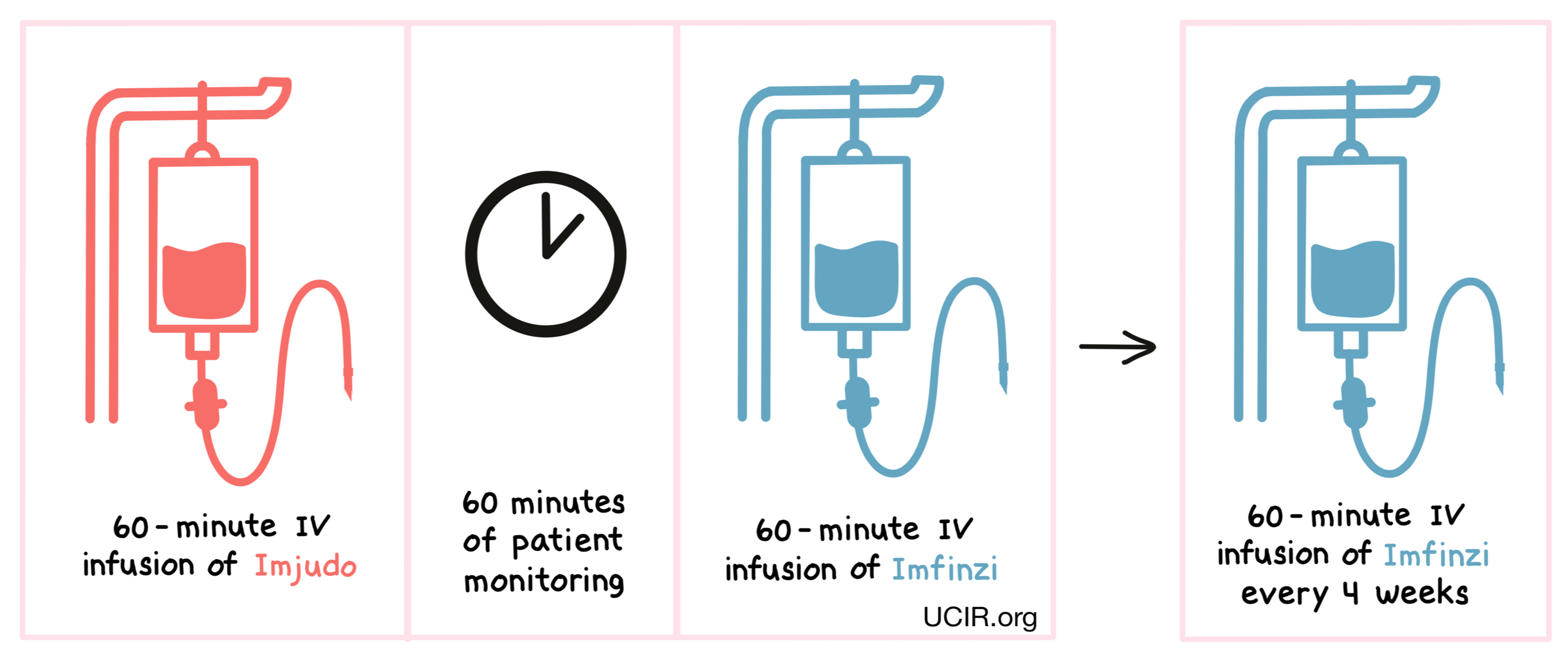
Imjudo is administered via a tube into a vein (intravenous infusion, or i.v.) over 60 minutes. Patients are then monitored for 60 minutes before receiving durvalumab (Imfinzi) via a tube in the vein over 60 minutes. For patients with liver cancer, therapy is then continued with only Imfinzi, administered every 4 weeks. Administration of Imjudo and Imfinzi does not require a hospital stay.
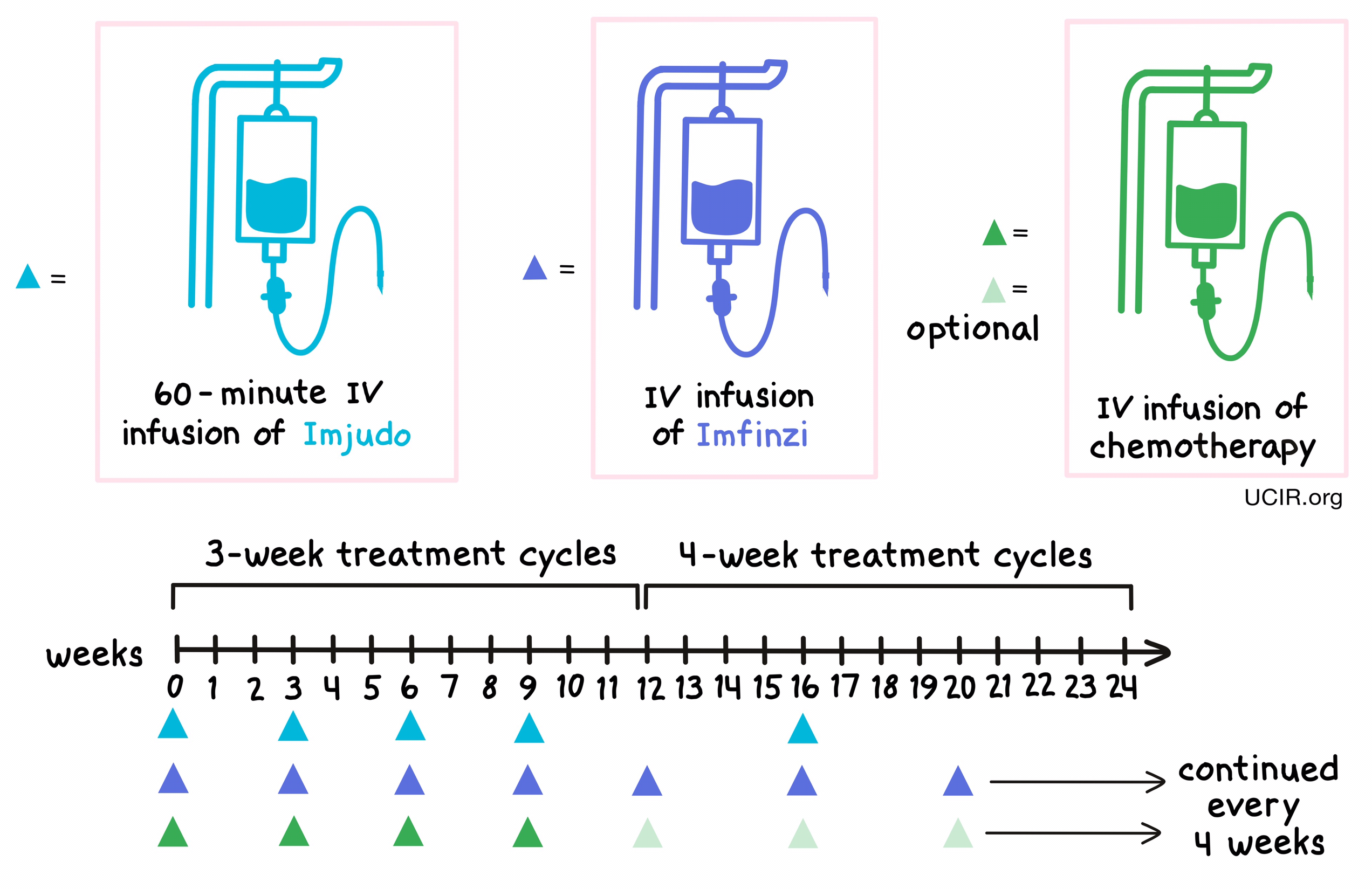
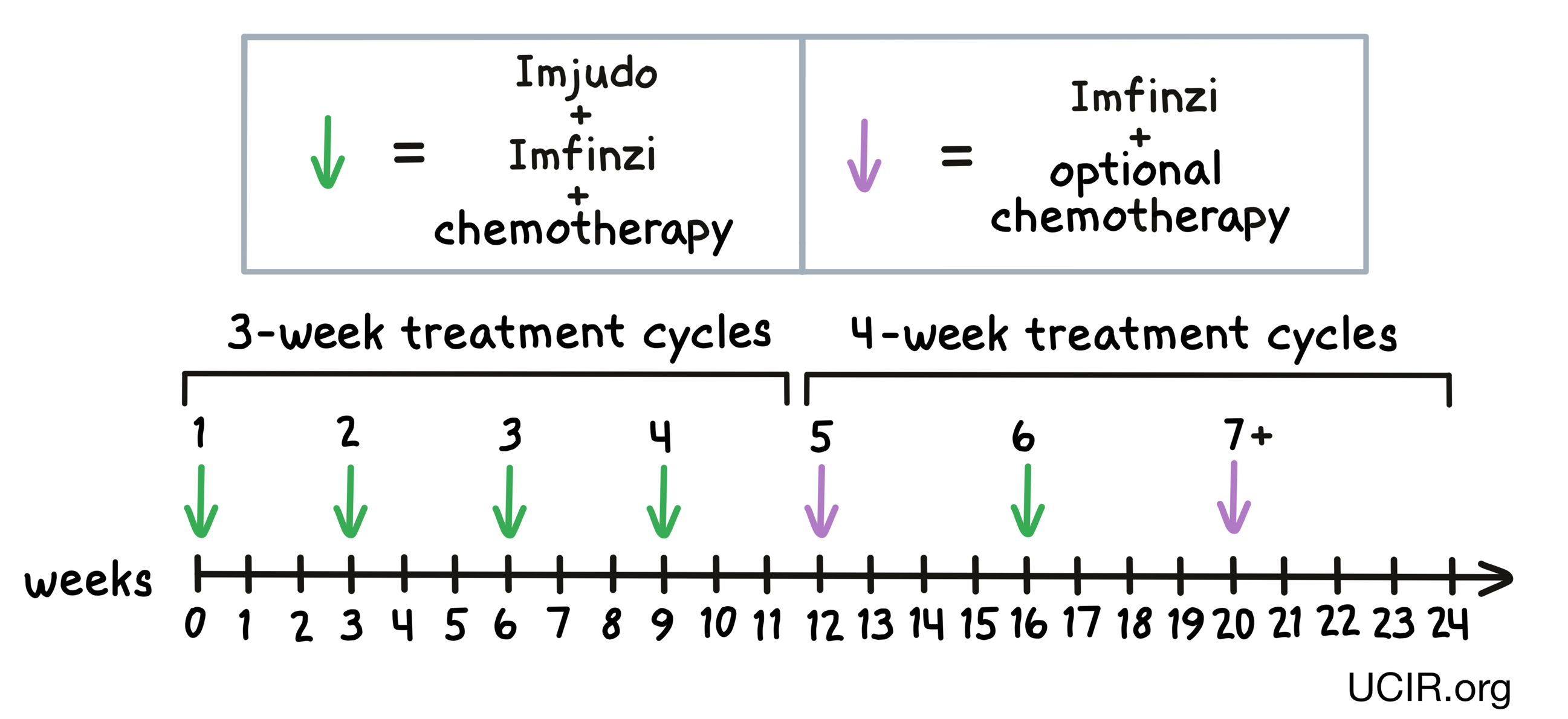
For patients with non-small cell lung cancer, Imjudo, durvalumab (Imfinzi), and platinum-based chemotherapy are given together (on the same day) at the beginning of each treatment cycle for treatment cycles 1 to 4 and treatment cycle 6. For the fifth treatment cycle, and for treatment cycles 7 and on, patients are treated only with Imfinzi and optional chemotherapy. Treatment cycles 1 through 4 are each 3 weeks long, while treatment cycles 5 and on are 4 weeks long.
What are the observed clinical results?
It is important to keep in mind that each patient’s actual outcome is individual and may be different from the results found in the clinical studies. In addition, with immunotherapy, sometimes it takes several months for responses to be observed.
Liver cancer
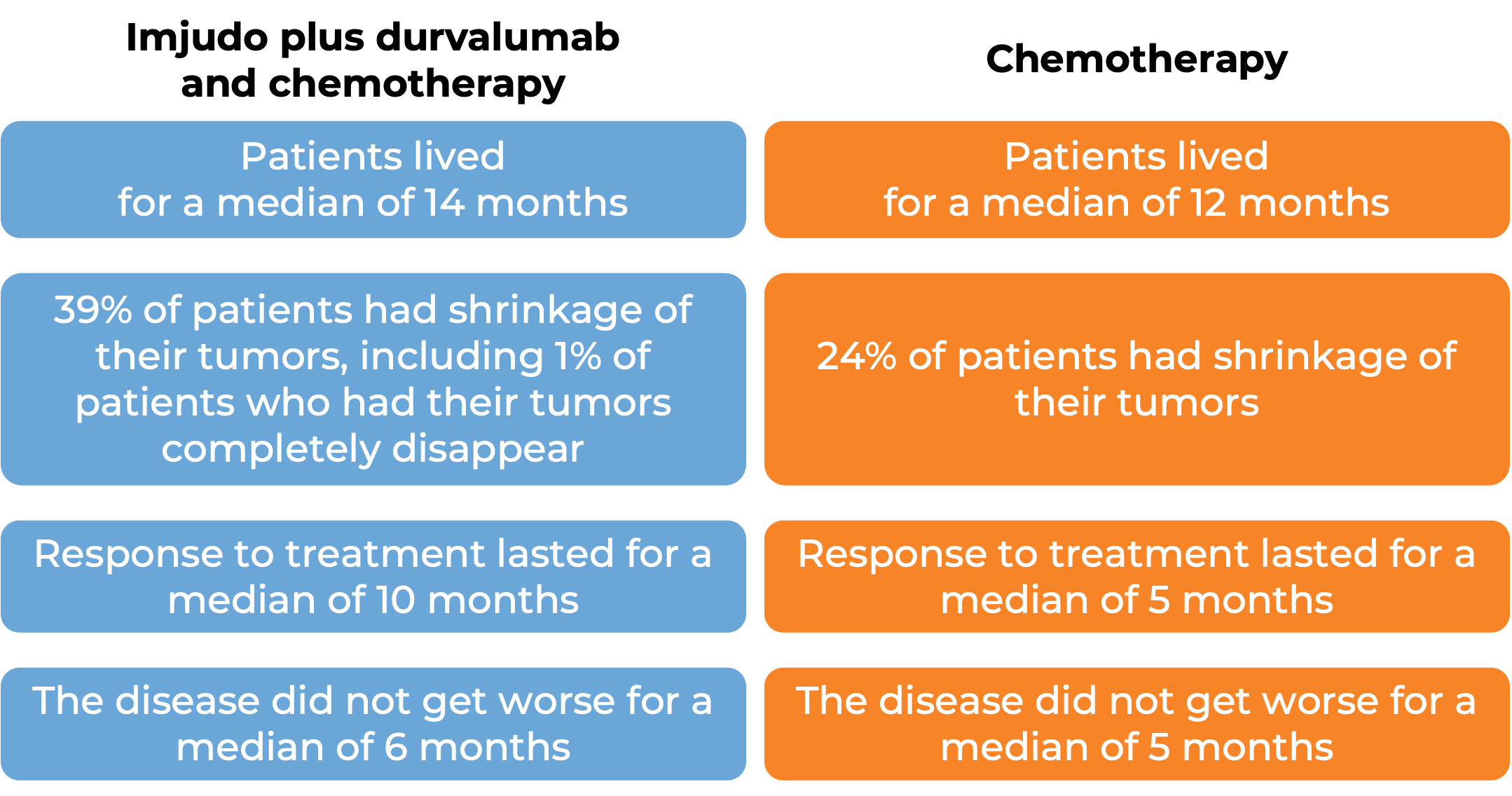
In a clinical study, 783 patients with liver cancer that cannot be removed by surgery were treated with Imjudo in combination with durvalumab (Imfinzi) or with sorafenib (Nexavar). At a median follow-up of 16 months for the Imjudo plus durvalumab treatment group and 13 months for the sorafenib treatment group:
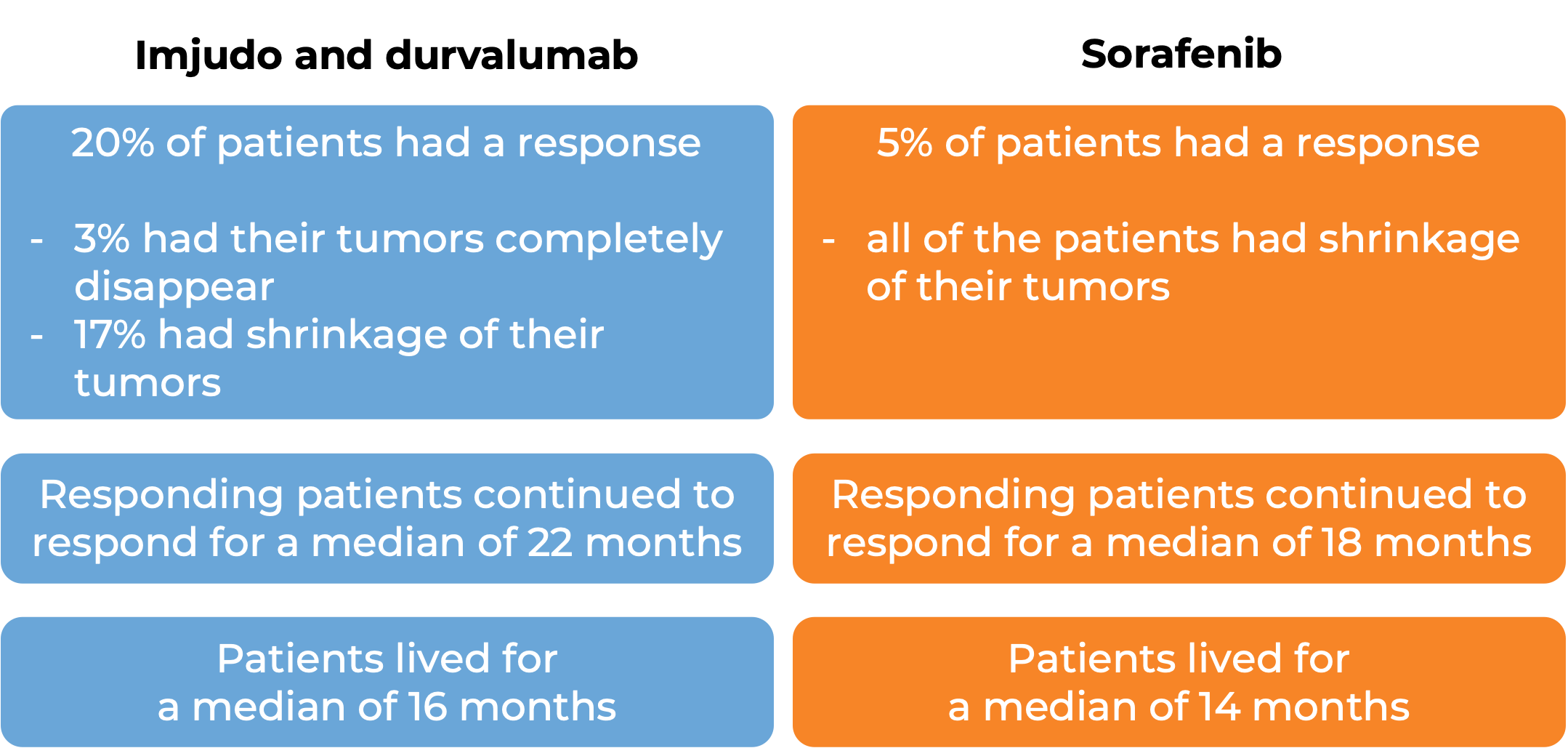
(For the definition of “median”, click here.)
Non-small cell lung cancer
In a clinical trial, 675 patients with non-small cell lung cancer that had spread to other parts of the body and did not have an abnormal EGFR or ALK gene, were treated with Imjudo, durvalumab (Imfinzi), and platinum-based chemotherapy, or platinum-based chemotherapy alone.
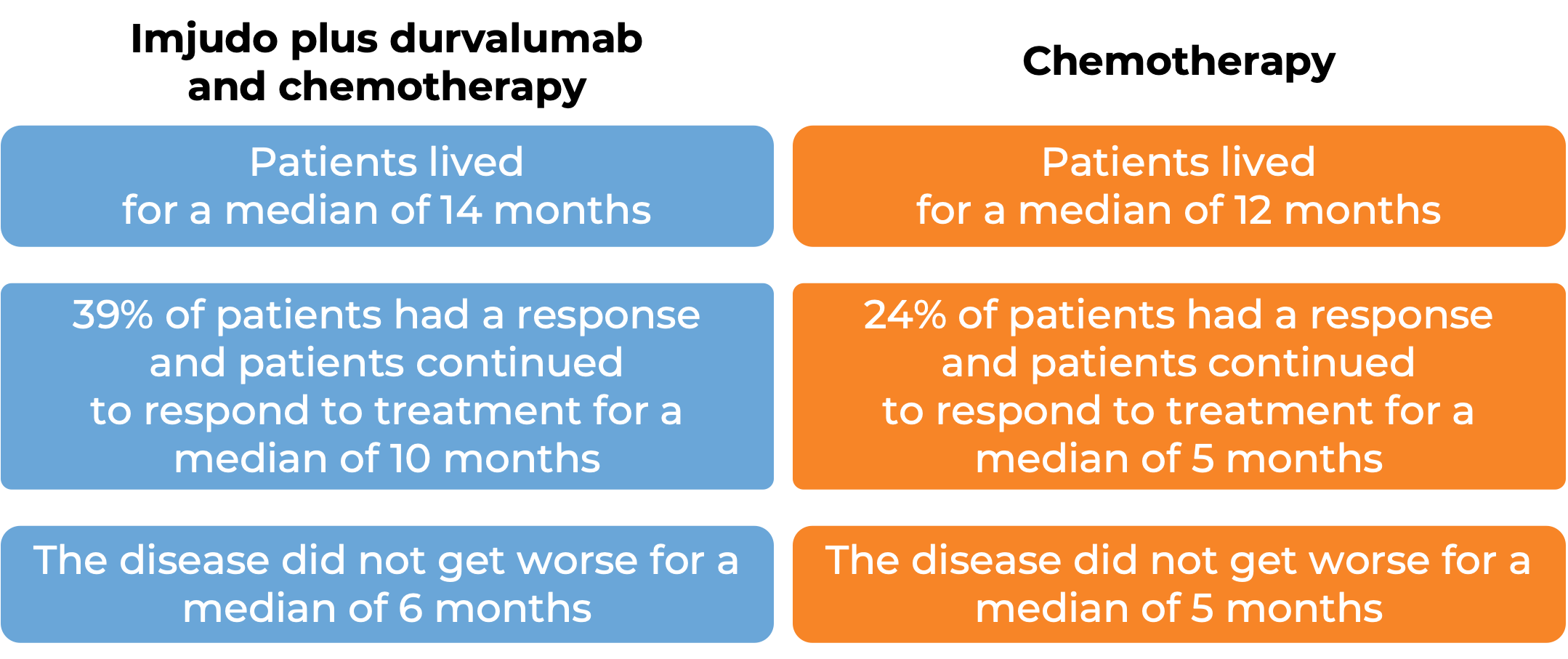
(For the definition of “median”, click here.)
What are the side effects?
The most common side effects of Imjudo when used in combination with durvalumab (Imfinzi) include fatigue, diarrhea, belly ache, itching, rash, and pain in the muscles, bones, nerves, ligaments, and tendons. The most common side effects of Imjudo when used in combination with durvalumab and platinum-containing chemotherapy are nausea, feeling tired or weak, pain in the muscles or bones, decreased appetite, rash, and diarrhea.
Imjudo can cause the patient’s T cells to attack healthy cells throughout the body. Because of this, Imjudo can cause side effects that may involve any organ, can become serious or life-threatening, and may lead to death. Some of the serious side effects related to Imjudo include inflammation of the lungs, brain, liver (which can lead to liver failure), kidneys (which can lead to kidney failure), or colon (which can result in tears or holes in the intestine). Additionally, problems can arise with hormone glands (including thyroid, pituitary, and adrenal glands, as well as the pancreas), and nerves (which can lead to paralysis). Skin rash (which could become severe and life-threatening) and reactions related to the infusion may also occur.
Patients should report any symptoms to their healthcare provider, who can then initiate actions to limit or reverse the side effects. For a more complete list of possible side effects, see the full prescribing information.
Additional information
Manufacturer
AstraZeneca
Approval
FDA and EMA
Links to drug websites
Other references
- Tremelimumab plus Durvalumab in Unresectable Hepatocellular Carcinoma. Abou-Alfa GK et al. The New England Journal of Medicine Evidence (2022)
- Phase 3 randomized, open-label, multicenter study of tremelimumab (T) and durvalumab (D) as first-line therapy in patients (PTs) with unresectable hepatocellular carcinoma (uHCC): HIMALAYA. Abou-Alfa GK et al. Meeting Abstract 2022 ASCO Gastrointestinal Cancers Symposium
Last updated: August 11, 2025